What are the valence electrons of Zn?
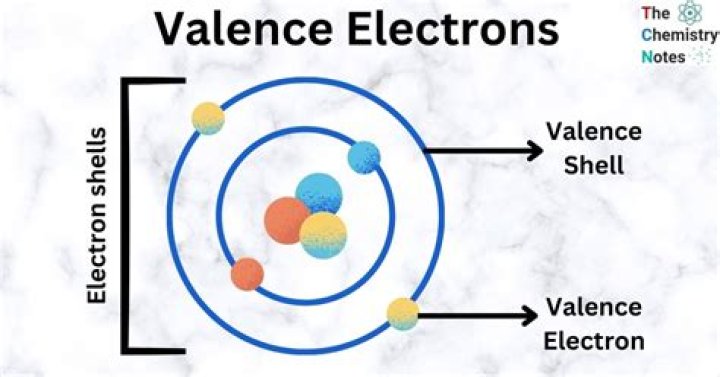
The electrons in the outermost or highest energy level orbitals. Hence, valence electrons in zinc are 2.
What color is zinc?
A silvery-white metal with a blue tinge. It tarnishes in air. Most zinc is used to galvanise other metals, such as iron, to prevent rusting.
How is zinc used?
Zinc uses range from metal products to rubber and medicines. About three-fourths of zinc used is consumed as metal, mainly as a coating to protect iron and steel from corrosion (galvanized metal), as alloying metal to make bronze and brass, as zinc-based die casting alloy, and as rolled zinc.
Where is zinc found?
Zinc is mined in over 50 countries with Canada being the leading producer, followed by Russia, Australia, Peru, United States and China. Deposits of zinc-bearing ores are found in most provinces of Canada, as well as in the Yukon and North West Territories.
How do you find valence electrons for zinc?
The atomic number zinc is 30 so, its electronic configuration is 1s22s22p63s23p63d104s2. The last shell is 4 and the electrons in the last shell are 2. So, the valence electrons of zinc are 2.
How many electrons are in zinc?
Zinc is located in period 4, group 12 of the periodic table and has an atomic number equal to 30. This means that neutral zinc atom has a total of 30 electrons surrounding its nucleus.
How many energy shells does zinc have?
Zinc atoms have 30 electrons and the shell structure is 2.8. 18.2. The ground state electron configuration of ground state gaseous neutral zinc is [Ar].
How do you write electronic configuration?
Writing Electron Configurations. When writing an electron configuration, first write the energy level (the period), then the subshell to be filled and the superscript, which is the number of electrons in that subshell. The total number of electrons is the atomic number, Z.
What is the electronic configuration of Zn and P?
The atomic number of zinc is 30, so 30 electrons are present in its neutral state, so configuration in neutral state will be: Zn =$1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{10}}4{{s}^{2}}$ , this is the electronic configuration of Zinc.
Is zinc an atom or molecule?
Zinc is a chemical element with symbol Zn and atomic number 30. Classified as a transition metal, Zinc is a solid at room temperature.
Is zinc element or compound?
zinc (Zn), chemical element, a low-melting metal of Group 12 (IIb, or zinc group) of the periodic table, that is essential to life and is one of the most widely used metals.
Where Does zinc come from?
Oysters contain more zinc per serving than any other food, but red meat and poultry provide the majority of zinc in the American diet. Other good food sources include beans, nuts, certain types of seafood (such as crab and lobster), whole grains, fortified breakfast cereals, and dairy products [2,11].
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
