What Bonds does cobalt make?
Cobalt also bonds with chlorine to form cobalt chloride and oxygen to form cobalt oxide. Cobalt oxide is particularly valuable and common as it is the cobalt complex that is used to provide a blue pigment to glassware that is otherwise difficult to synthesize.
What can cobalt bond with?
Cobalt is one of the three metals that are ferromagnetic at room temperature. It dissolves slowly in dilute mineral acids, does not combine directly with either hydrogen or nitrogen, but will combine, on heating, with carbon, phosphorus, or sulfur.
What compounds does cobalt make?
Cobalt compounds are commonly used to make colored glass, glazes, paints, rubber, inks, cosmetics, and pottery. These compounds compounds include: cobalt oxide, cobalt potassium nitrite, cobalt aluminate, and cobalt ammonium phosphate. Cobalt compounds can also be used as catalyst.
Cobalt sulfide is the name for chemical compounds with a formula CoxSy. Well-characterized species include minerals with the formula CoS, CoS2, Co3S4, and Co9S8. In general, the sulfides of cobalt are black, semiconducting, insoluble in water, and nonstoichiometric.
How many covalent bonds can cobalt form?
(b) The cobalt(III) complex [Co(en)3]3+ contains three of these ligands, each forming two bonds to the cobalt ion. Any ligand that bonds to a central metal ion by more than one donor atom is a polydentate ligand (or “many teeth”), because it can bite into the metal center with more than one bond.
Cobalt and bromine, ΔEN = 0.9, polar covalent.
Why is cobalt a transition element?
The 38 elements in groups 3 through 12 of the periodic table are called “transition metals”. As with all metals, the transition elements are both ductile and malleable, and conduct electricity and heat. These elements are iron, cobalt, and nickel, and they are the only elements known to produce a magnetic field.
What are chemical properties of cobalt?
Properties Of Cobalt
It is a hard ferromagnetic, silver-white, lustrous, brittle element.It is stable in air and does not react with water.Like other metals, it can also be magnetized.With dilute acids, it reacts slowly.The metal melts at 1495 °C and boils at 2927 °C.
The period 4 transition metals are scandium (Sc), titanium (Ti), vanadium (V), chromium (Cr), manganese (Mn), iron (Fe), cobalt (Co), nickel (Ni), copper (Cu), and zinc (Zn).
How is cobalt formed in nature?
Most cobalt is formed as a by-product of nickel refining. A huge reserve of several transition metals (including cobalt) can be found in strange nodules on the floors of the deepest oceans. The nodules are manganese minerals that take millions of years to form, and together they contain many tonnes of cobalt.
What is cobalt used for?
Cobalt is also used to make airbags in automobiles; catalysts for the petroleum and chemical industries; cemented carbides (also called hardmetals) and diamond tools; corrosion- and wear-resistant alloys; drying agents for paints, varnishes, and inks; dyes and pigments; ground coats for porcelain enamels; high-speed
What is the element of co?
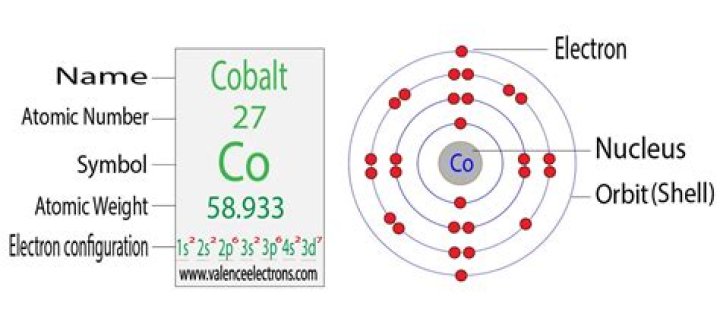
Cobalt is a chemical element with the symbol Co and atomic number 27. Like nickel, cobalt is found in the Earth’s crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron.
Atoms bond by sharing electrons. In a typical bond two electrons are shared, one from each of the atoms involved. Carbon has four such sharable electrons of its own, so it tends to form four bonds to other atoms.
How many protons does cobalt have?
Cobalt-60 has 27 protons (thus 33 neutrons). If an inter-nucleus neutron decays and emits an electron, this neutron transforms into a proton. Now we have 28 protons and 32 neutrons.
How covalent bonds are formed?
A covalent bond forms when the difference between the electronegativities of two atoms is too small for an electron transfer to occur to form ions. Shared electrons located in the space between the two nuclei are called bonding electrons. The bonded pair is the “glue” that holds the atoms together in molecular units.
What type of bond is involved in the formation of MgCl2?
MgCl2 is an ionic compound because the bond formed between magnesium and the chlorine atom is ionic in nature, it is formed as magnesium atom loses two electrons to form Mg2+ ion and each chlorine accepts the one electron to form Cl– ion, later these ions(Mg2+ and 2Cl–) are attracted by the electrostatic force of
Silicon dioxide is a covalent compound, rather it is a “polar covalent compound”, in which each silicon atom of a crystal is covalently bonded with four atoms of oxygen and each oxygen is bonded with two silicon atoms.
What kind of element usually forms covalent bond?
Covalent bonds usually occur between nonmetals. For example, in water (H2O) each hydrogen (H) and oxygen (O) share a pair of electrons to make a molecule of two hydrogen atoms single bonded to a single oxygen atom. In general, ionic bonds occur between elements that are far apart on the periodic table.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
