What causes a dipole dipole force?
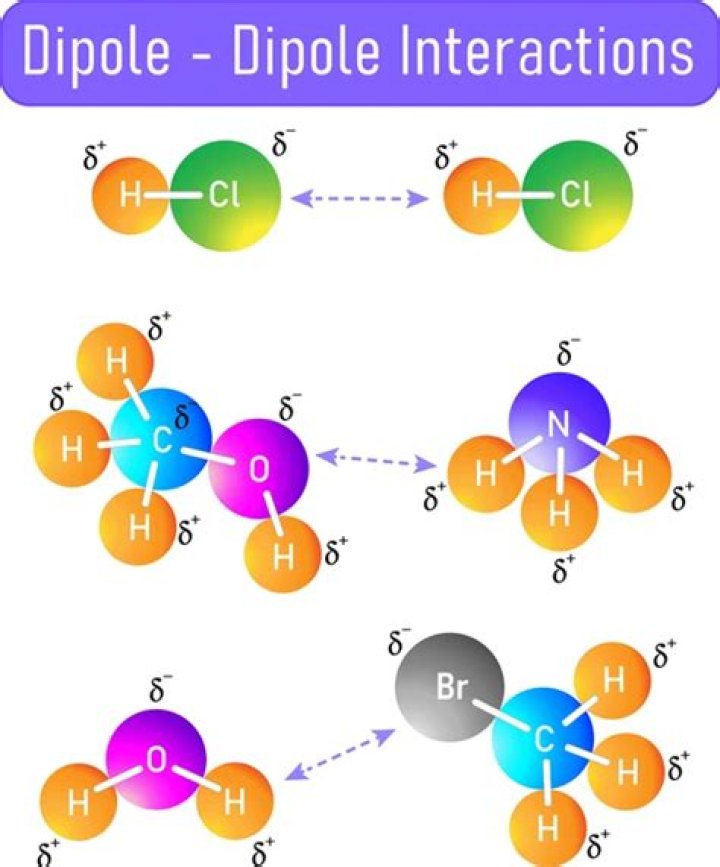
Dipole-dipole interactions occur when partial charge form within a molecule because of the uneven distribution of electrons. Polar molecules align so that the positive end of one molecule interacts with the negative end of another molecule.
What are dipole-dipole forces called?
Instantaneous dipole-induced dipole forces or London dispersion forces: forces caused by correlated movements of the electrons in interacting molecules, which are the weakest of intermolecular forces and are categorized as van der Waals forces.
What do intramolecular forces do?
An intramolecular force (or primary forces) is any force that binds together the atoms making up a molecule or compound, not to be confused with intermolecular forces, which are the forces present between molecules.
Why are dispersion forces high in molecules?
Larger and heavier atoms and molecules exhibit stronger dispersion forces than smaller and lighter ones. In a larger atom or molecule, the valence electrons are, on average, farther from the nuclei than in a smaller atom or molecule. They are less tightly held and can more easily form temporary dipoles.
What is dipole-dipole forces simple?
Dipole-dipole forces are attractive forces between the positive end of one polar molecule and the negative end of another polar molecule. They are much weaker than ionic or covalent bonds and have a significant effect only when the molecules involved are close together (touching or almost touching).
What are ion-dipole forces quizlet?
ion-dipole forces. attractive forces between an ion and a polar molecule. – ions can interact with partial forces/charges. London dispersion forces. The intermolecular attractions resulting from the constant motion of electrons and the creation of instantaneous dipoles.
What is the ion-dipole force Why is it important quizlet?
The ion-dipole force occurs when an ionic compound is mixed with a polar compound. It is especially important in aqueous solutions of ionic compounds. They are the strongest IMF. They are responsible for the ability of ionic substances to form solutions with water.
What affects the strength of dipole-dipole forces?
Larger atoms more electrons to slosh around easier to polarize. Which Have a Greater Effect? If two molecules are of comparable size and shape, dipole–dipole interactions will likely be the dominating force. If one molecule is much larger than another, dispersion forces will likely determine its physical properties.
How are dispersion forces different to dipole-dipole interactions?
The main difference between dipole-dipole and London dispersion forces is that dipole-dipole forces occur among molecules with dipole moment whereas London dispersions occur due to instantaneous dipoles that form in atoms or nonpolar molecules.
What is the strongest intermolecular force?
Dipole-dipole interactions are the strongest intermolecular force of attraction.
How might dipole-dipole interactions help many molecules attract each other?
How might dipole-dipole interactions help many molecules attract each other? Dipoles are held together by the attraction between the oppositely charged ends of the separate molecules for each other.
Which of the following interaction is dipole induced dipole?
The correct answer is (B) HCl and He Atoms. The forces or interactions, which occur between a polar molecule and a nonpolar molecule or with an atom are termed as Dipole -induced dipole interactions. Here, HCl is polar while helium is non-polar; thus, it is an example of Dipole induced dipole.
Which force holds molecules together?
An intermolecular force (IMF) (or secondary force) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction or repulsion which act between atoms and other types of neighboring particles, e.g. atoms or ions.
Why are intramolecular forces stronger than intermolecular forces?
Intramolecular force is stronger than intermolecular force. Intramolecular force is due to strong covalent bond or ionic bond. On the other hand intermolecular forces are very quick forces like London force,van dar wall force etc.
Why are intermolecular forces different from intramolecular forces?
Intramolecular forces are forces that occur within a molecule, while intermolecular forces are forces that occur between molecules. Intermolecular forces prevent each individual molecule from leaving the other molecules. In water, the intermolecular force of attraction is due to: hydrogen bonding.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
