What does versatility of a solvent mean?
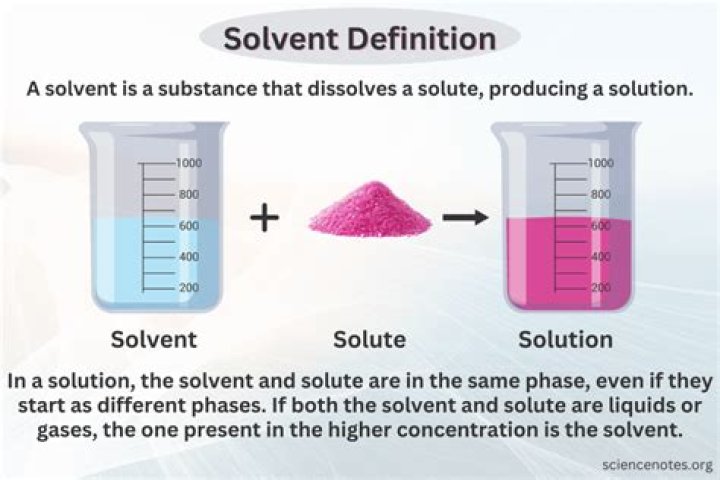
Solution – A liquid that is a homogenous mixture of two or more substances.
What is versatility as a solvent in water?
Water is versatile because of its polarity, which allows it to form hydrogen bonds easily with certain solutes. What ionic compounds dissolve in water? Some ionic compounds like salt dissolve in water because the ions become separated by water molecules, thus dissolving.
How is versatility as a solvent important to life?
Water is called the “universal solvent” because it is capable of dissolving more substances than any other liquid. This is important to every living thing on earth. It means that wherever water goes, either through the air, the ground, or through our bodies, it takes along valuable chemicals, minerals, and nutrients.
What is the most versatile solvent?
Water is often called the universal solvent because it dissolves more chemicals than any other solvent.
What would happen if water was not a versatile solvent?
What would happen if water didn’t act as a universal solvent? – Quora. had water not been the universal solvent then day to day food stuff like salt in water or sugar in water would never be able to dissolve. this is at domestic level. at a larger view separation of chemicals would be very difficult .
What does universal solvent mean?
From Wikipedia, the free encyclopedia. Universal solvent may refer to: Water, described as the “universal solvent” for its ability to dissolve many substances. Alkahest, a hypothetical solvent able to dissolve every other substance.
Do all solutes dissolve in solvents?
No. Generally both solvents and solutes are separated into two types: polar/ionic and non-polar. The rule of thumb is that “like dissolves like”. Polar/ionic solvents dissolve polar/ionic solutes and non-polar solvents dissolve non-polar solutes.
How does the ability of water to function as a versatile solvent quizlet?
Water’s polarity causes it to be a versatile solvent. The slightly negative charged oxygen and slightly positive charged hydrogen atoms are attracted to the dissolved molecule. Water creates a hydration shell around ionic compounds, dissociating the initial ionic bond of the solute.
What dissolves in water list?
Expect the following results.
Salt. Will dissolve (disappear), leaving a clear solution.Sugar: Will dissolve (disappear), leaving a clear solution.Flour. Oil. Food coloring. Coffee.
What is the solvent in a cup of milk?
Milk has: water, proteins, fats, lactose, minerals, and vitamins. Among these: Lactose, some minerals, water soluble vitamins are dissolved in water. So they can be considered as solutes and water is their solvent.
Is oil a universal solvent?
A true universal solvent does not exist. Water is often called the universal solvent because it dissolves more chemicals than any other solvent. However, water only dissolves other polar molecules. It does not dissolve nonpolar molecules, including organic compounds such as fats and oils.
Why water is known as a universal solvent?
Water is called the universal solvent because more substances dissolve in water than in any other chemical. This has to do with the polarity of each water molecule. The hydrogen side of each water (H2O) molecule carries a slight positive electric charge, while the oxygen side carries a slight negative electric charge.
Is water considered a universal solvent?
Water as the “Universal Solvent”
Because of water’s polarity, it is able to dissolve or dissociate many particles.
How does the ability of water to function as a versatile solvent arise from the structure of water molecules?
Technically speaking water is not the universal solvent however it’s make up allows other molecules to “dissolve”. Water uses H-Bonds and polar covalent molecules to dissolve other polar molecules. The opposite ends of the water molecules attract to each other and form hydrogen bonds.
What is being dissolved in a solution?
The substance being dissolved is called the solute and the liquid doing the dissolving is called the solvent.
Is sugar a solute or solvent?
The solvent is the component that dissolves the other component. Solute refers to the component that are dissolved in the solvent. For a sugar solution, the solute will be sugar and solvent will be water.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
