What is difference between density and specific gravity?
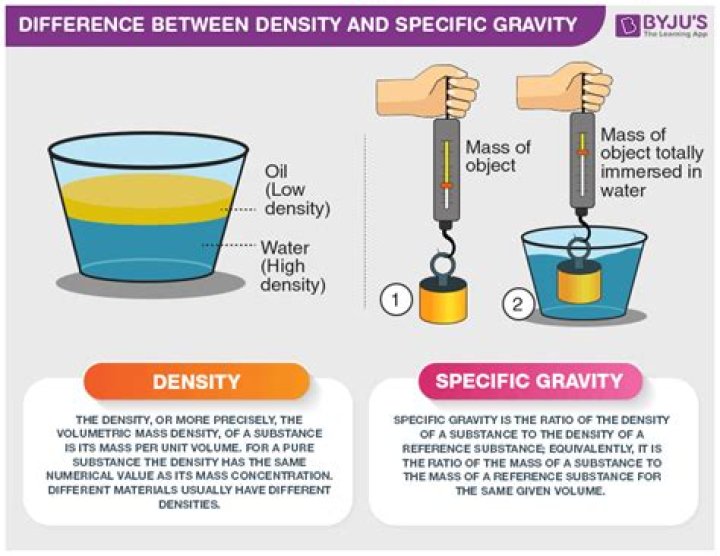
Density is defined as mass per unit volume. It has the SI unit kg m-3 or kg/m3 and is an absolute quantity. Specific gravity is the ratio of a material’s density with that of water at 4 °C (where it is most dense and is taken to have the value 999.974 kg m-3). It is therefore a relative quantity with no units.
What is the difference between density and specific gravity quizlet?
What is the difference between density and specific gravity? Density is the relations hip of a substance’s mass to its volume and has specific units whereas specific gravity refers to the comparison of a substance’s density with that of water’s and has no specific units.
How do you convert density to specific gravity?
Converting a specific gravity into a density is as simple as multiply the SG by the reference density for which it was based. In most cases, this is the density of water, and therefore your multiply the SG by 1 to yield your density in g/cm^3.
Specific gravity, also called relative density, ratio of the density of a substance to that of a standard substance. The usual standard of comparison for solids and liquids is water at 4 °C (39.2 °F), which has a density of 1.0 kg per litre (62.4 pounds per cubic foot).
Is specific gravity g mL?
The specific gravity of an object is the ratio between the density of an object to a reference liquid. Usually, our reference liquid is water, which has a density of 1 g/mL or 1 g/cm^3. The specific gravity has no units.
What is the difference between density and bulk density? – Density is a concept defined for any substance, while bulk density is only used in cases where the particles or chunks of matter are loosely packed with space for air within. – For normal solids and liquids the bulk density and density are the same.
What is SI unit of density?
The formula for density is d = M/V, where d is density, M is mass, and V is volume. Density is commonly expressed in units of grams per cubic centimetre. Density can also be expressed as kilograms per cubic metre (in metre-kilogram-second or SI units).
What’s the difference between specific weight and density?
The density of the material is defined as mass per unit volume, typically measured in kg/m3. The standard gravity is acceleration due to gravity, usually given in m/s2, and on Earth usually taken as 9.81 m/s2. Unlike density, specific weight is not a fixed property of a material.
You are asked to calculate density. Hence, you take the formula for density, which is d = m / V. Then, you plug into the formula the values of mass and volume and calculate. d = 40.5 g / 15.0 mL = 2.70 g/mL A zero after 2.7 is added as you need 3 significant figures.
What is the relationship of density to specific gravity?
Density = Mass / Volume
Specific gravity is the density of a substance divided by the density of water. Since (at standard temperature and pressure) water has a density of 1 gram/cm3, and since all of the units cancel, specific gravity is usually very close to the same value as density (but without any units).
What is the difference between density and mass quizlet?
What is the difference between density and mass? The density of a substance is independent of the amount of the substance, while mass depends on the amount of the substance.
Why specific gravity test is done?
A urine specific gravity test compares the density of urine to the density of water. This quick test can help determine how well your kidneys are diluting your urine. Urine that’s too concentrated could mean that your kidneys aren’t functioning properly or that you aren’t drinking enough water.
As per the specific gravity definition, the value is calculated as the density of that substance divided by water density at 4⁰C. To obtain the specific weight of a substance, its specific gravity is multiplied by 1000 (density of water in kg/m³).
What’s the specific gravity of water?
In less dense liquids the hydrometer will float lower, while in more dense liquids it will float higher. Since water is the “standard” by which other liquids are measured, the mark for water is probably labeled as “1.000”; hence, the specific gravity of water at about 4°C is 1.000.
What is difference between density and relative density?
Density is the ratio between the mass and the volume of a body. Relative density, on the other hand, is the ratio between the density of an object (substance) and the density of some other reference object (substance) at some given temperature. Density is measured in, while relative density is dimensionless.
What is the difference between specific density and relative density?
Density is the property of matter represented by a ratio of mass to a unit volume of matter. Specific gravity which is also called as relative density is a measure of density with respect to a density of pure water.
Since the density of a liquid varies as its temperature changes, the scale is adjusted to a certain temperature, usually about 15 degrees C., at which determinations must be made. Its scale is adapted to liquids heavier or lighter than water.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
