What is formed when sugar as C6H12O6 is dissolved in water?
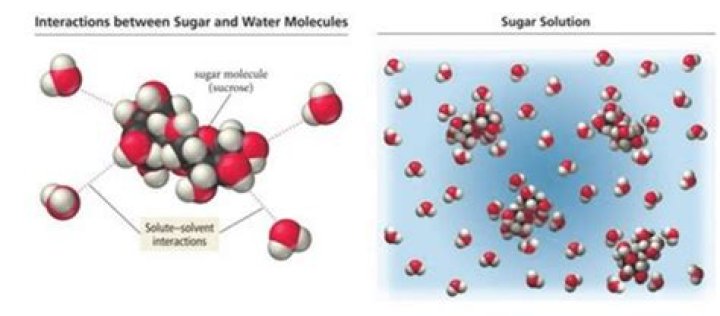
Because C6H12O6 (glucose) is a covalent compound, it is formed of molecules and stays as molecules when dissolved in water. In other words, it does not dissociate into ions as an ionic compound like salt does (NaCl → Na+ + Cl-).
What is formed when sugar as C6H12O6 is dissolved in water?
C6H12O6 is the chemical formula of glucose. Reaction : C6H12O6 (s) + H2O (l) 鈫扖6H12O6 (aq) If we dissolved any substance in water,it would be formed into it’s aqueous solution.
Why is C6H12O6 soluble in water?
(a) The structures for glucose, C6H12O6 , and cyclohexane, C6H12 , are shown below. The hydroxyl groups in glucose molecules can form strong hydrogen bonds with the solvent (water) molecules, so glucose is soluble in water.
What happens when glucose reacts water?
Glucose can be reacted in water to obtain yields of 5-(hydroxymethyl)furfural (5-HMF) as high as 27% at 180 掳C in 3 min reaction time [39], and when glucose is reacted in ionic liquid solvents with CrCl2, 5-HMF yields of 70% can be obtained at 100 掳C in 3 h [40]. Kabyemela et al.
What is the dissociation equation for C6H12O6?
The breakdown of glucose living organisms utilize to produce energy is described by the equation: C6H12O6+6O2鈫6CO2+6H2O+energy C 6 H 12 O 6 + 6 O 2 鈫 6 CO 2 + 6 H 2 O + energy .
What is sugar C6H12O6?
Glucose is a simple sugar with the molecular formula C6H12O6. Glucose is the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is a monosaccharide containing six carbon atoms and an aldehyde group, and is therefore an aldohexose.
Is dissociation the same as solubility?
Solubility is said to be saturated when the maximum amount of solute is dissolved in the solvent at a given temperature. Dissociation occurs when an ionic compound is dissolved in water due to interactions between the charges and the polarity of atoms in the water molecule.
How does CaCl2 dissociate in water?
Calcium chloride when dissolved in water dissociates into its ions according to the following equation. CaCl2 (aq) 鈫 Ca2+ (aq) + 2Cl鈥 (aq) Calculate the number of ions obtained from CaCl2 when 222 g of it is dissolved in water.
What are the bonds in C6H12O6?
Yes, this compound is known as glucose (a sugar). a). This compound is made of C-H bonds, C-O bonds and O-H bonds. All of these form covalent bonds because they share electrons and the difference in electronegativity values aren’t great enough to form ionic bonds.
Is C6H12O6 solid or liquid?
It can occur either in the solid or liquid form. It is water-soluble and is also soluble in acetic acid. It is odourless and sweet to taste.
Is C6H12O6 polar or nonpolar?
The 鈥渦sual suspects鈥 for a molecule with formula C6H12O6 鈥 glucose, fructose, galactose 鈥 are polar molecules, and as a result dissolve easily in water.
Which compound is the most soluble in water?
From the given compounds phenol is most soluble in water due to the following reasons.
It is slightly polar.It can form hydrogen bonds with
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
