What is Group 13 on the periodic table called?
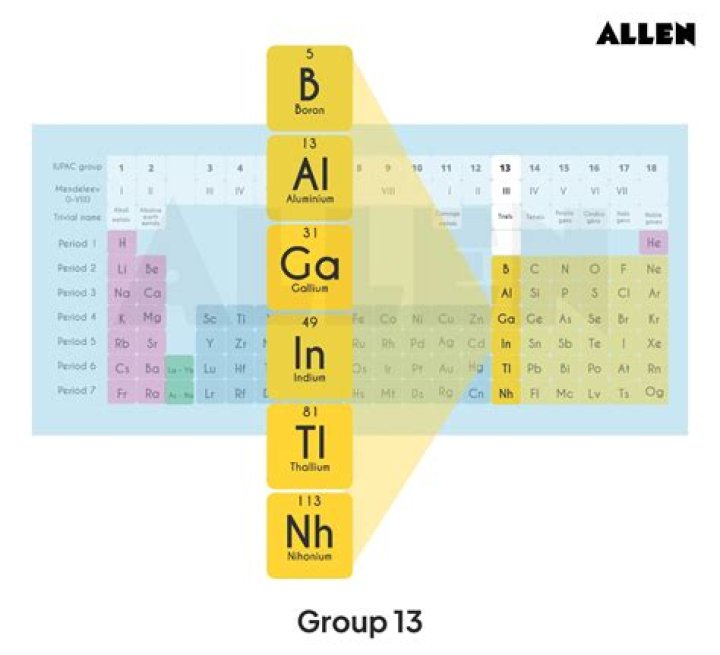
boron group element, any of the six chemical elements constituting Group 13 (IIIa) of the periodic table. The elements are boron (B), aluminum (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh).
What is group 13 15 on the periodic table called?
Group 13 (Boron Group)
The Boron group is the series of elements in group 13 (formerly group III) in the periodic table. It consists of the elements boron (B), aluminium (Al), gallium (Ga), indium (In), thallium (Tl), and ununtrium (Uut) (unconfirmed).
Are group 13 elements metals?
The boron family contains elements in group 13 of the periodic talbe and include the semi-metal boron (B) and the metals aluminum (Al), gallium (Ga), indium (In), and thallium (Tl). It is classified as a metalloid due it its properties that reflect a combination of both metals and nonmetals.
Indium and thallium, the heavier group 13 elements, are found as trace impurities in sulfide ores of zinc and lead. Indium is used as a crushable seal for high-vacuum cryogenic devices, and its alloys are used as low-melting solders in electronic circuit boards.
Which of the following is NOT group 13?
All the elements of group 13 are also called the boron family. The elements of the 13th group element are boron, aluminium, gallium, indium, and thallium. They all are metallic in nature except boron which is a metalloid.
It is highly reactive, and therefore found in nature in compounds. Aluminum does not appear to react with water because it is aluminum is protected by a layer of Al2O3; this effect is known as anodizing.
Which element of group 13 is non metallic?
The non-metal that is present in Group 13 of the periodic table the elements is Boron.
Is group 13 reactive?
Moreover, all of the other elements in group 13 are relatively reactive at moderate temperatures, while boron’s reactivity only becomes comparable at very high temperatures. One characteristic that all do have in common is having three electrons in their valence shells.
The small size of the boron 3+ means it has a very large charge to radius ratio meaning it is not stable. This forms covalent bonds with elements as the bonding is highly polarizing pulling the electron density away from the element.
What is Group 14 called?
carbon group element, any of the six chemical elements that make up Group 14 (IVa) of the periodic table—namely, carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl).
What are the 13 elements of nature?
The twelve elements of nature are Earth, Water, Wind, Fire, Thunder, Ice, Force, Time, Flower, Shadow, Light and Moon. Each of these elements are simplified terms for higher and complex substances.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
