What is the best structure for N2O?
Structure 3 is the best (most stable) structure we can draw for N2O. It’s center atom contains, around it, two sigma (σ) bonds.
What geometric shape is n2o?
The molecular geometry of N2O is linear.
What is the correct electron dot structure of n2o?
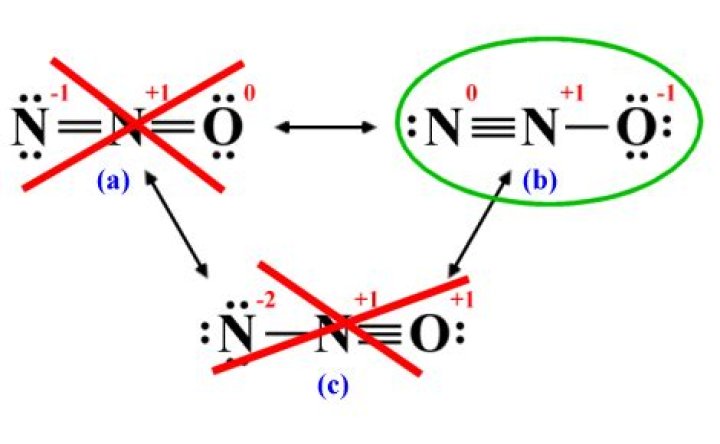
The given molecule is,
The nitrogen has ‘5’ valence electrons and oxygen has ‘6’ valence electron. According to electron-dot structure, there are 8 number of bonding electrons and 8 number of non-bonding electrons. In the structure of , one N molecule act as a central atom and is bonded to another N and O atom.
Dinitrogen monooxide, or N2O , has three resonance structures, out of which one is a major contributor and one is a minor contributor. Moreover, it shows a positive charge placed on the more electronegative oxygen atom and a very destabilizing (-2) formal charge on a nitrogen atom.
What type of intermolecular force is N2O?
N2O is a neutral molecule, nitrous oxide, it is isoelectronic with CO2. The intermolecular force which polar molecules take part in are dipole-dipole forces.
The N2O molecule is linear and polar.
Which of the following resonating structures of N2O is the most contributing structure?
In N2O molecule N=N-O structureis most contributed.
Which resonance structure contributes the most to the overall structure of SCN − SCN −?
Structure #1 is the most stable resonance Lewis structure since the octet rule is obeyed and the negative formal charge is carried out by N (electronegativity: 3.04) the most electronegative atom compared to S (electronegativity: 2.58).
Since the right nitrogen “owns” four valence electrons (one lone pair gives two electrons; then, two electrons from the N=O. double bond), and it expects five, its formal charge is 5−4=+1 .
How do you choose a good resonance structure?
Using Formal Charges to Identify viable Resonance Structures
The greater the number of covalent bonds, the greater the stability since more atoms will have complete octets.The structure with the least number of formal charges is more stable.The structure with the least separation of formal charge is more stable.
Why is N2O linear?
In the diagram given above, the type of bonding in nitrous oxide is covalent bonding. Due to the presence of sigma bonds and absence of lone pairs on the central atom, the stable structure of nitrous acid is linear. It is also known as sulphur dioxide.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
