What is the bond between P4?
In P4, all of the atoms bonding are the same. There will be no polarity between in the bonds between the atoms, and thus no polarity in the compound as a whole. Therefore, tetraphosphorus is nonpolar since each phosphorus atom’s electron pull in opposing directions is the same and thus cancel each other out.
What is the bond of P4?
The Phosphorus P4 molecule contains a total of 3 bond(s) There are 3 non-H bond(s), 3 multiple bond(s), 1 double bond(s) and 2 triple bond(s). The 2D chemical structure image of Phosphorus P4 is also called skeletal formula, which is the standard notation for organic molecules.
How many sigma bonds are there in P4?
Thus, the total number of sigma bonds for each phosphorus atom is three for the three [P – O] single bonds and one for the one [P = O] double bond, so total of four sigma bonds.
P4 is a phosphorus molecule. Here 4 is after the atomic symbol ( a subscript) whenever there is a subscript ( a number after the symbol of the element ) it means that the two or more then two atoms of the same element are in chemical bonding and in this case we call it a molecule.So, P4 is a phosphorus molecule.
What is the molecular structure of P4?
The shape of P4 is tetrahedral. Each P atom makes 3 bonds and has a lone pair, making each formal charge 0.
Structure of white phosphorous (P4) molecules as: Each P atom has one lone pair of electrons and each covalent bond has two electrons of a bond pair. In a white phosphorus molecule, there are four lone pairs of electrons on four phosphorus atoms and six P−P single bonds are present.
What is the total number of single bonds formed in P4 molecule?
White phosphorus (P4) has 6P−P single bonds, 4 lone pairs of electrons and P−P−P angle of 60∘.
Is Br2 covalent bond?
Br2 is a diatomic molecule having two bromine atoms is a linear-shaped structured compound. The atoms forming a covalent bond having equal electronegativity are nonpolar in nature. Therefore, the Br2 molecule is a nonpolar molecule.
P4 is a substance that is made up of four atoms of the same element, so it is a molecular element. The formula for oxygen gas is O2 so it is a molecular element.
How is p4 formed?
Answer: Phosphorus can form a P4 white phosphorus tetrahedron because it can form three bonds. P has a high atomic size and a low tendency for forming triple bonds. It can make a tetra-atomic P4 molecule by sharing valency electrons with three other P atoms to complete its octet.
What is the hybridization of p4?
Hybridization of Phosphorus in P4: It exists as molecules made up of four atoms in a tetrahedral structure. The tetrahedral arrangement results in ring strain and instability.
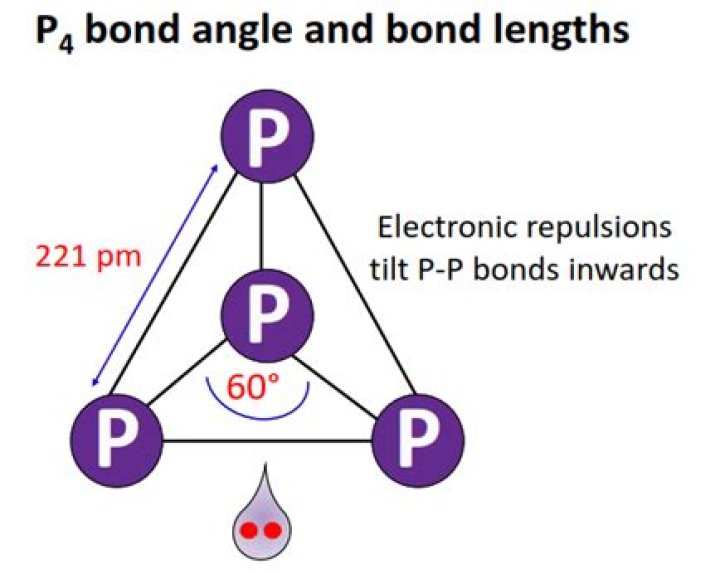
What is PPP bond angle P4?
The P-P-P bond angle in a tetrahedral P4 molecule is only 60o.
(b, d) Structure of P4 molecule can be represented as It has total four lone pairs of electrons situated at each P – atom.
How many P P single bonds are present in P4 molecule?
Therefore, in total there will be 4 lone pair of electrons and six P-P single bonds in a molecule of white phosphorus.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
