What is the charge an aluminum ion?
Please visit the Aluminum element page for information specific to the chemical element of the periodic table. Aluminium(3+) is an aluminium cation that has a charge of +3.
What is an aluminum ion?
Aluminium-ion batteries are a class of rechargeable battery in which aluminium ions provide energy by flowing from the positive electrode of the battery, the anode, to the negative electrode, the cathode. When recharging, aluminium ions return to the negative electrode, and can exchange three electrons per ion.
How do you find the ions of Aluminium?
Answer :1 mole of Aluminium Oxide (Al2O3) = 2 × 27 + 3 × 16 = 102 g. We know, The number of atoms present in given mass = (Given mass ÷ molar mass) × Avogadro number.Then, 0.051 g of Aluminium Oxide (Al2O3) contains.= (0.051 ÷ 102 ) × 6.022 × 1023 molecules of Aluminium Oxide (Al2O3)
The Lewis symbol for an aluminum ion, Al3+, is A.) Al atomic number is 13 which means it has 13 electrons and 13 protons.
What is the charge on the ion formed by aluminum quizlet?
An aluminum (Al) ion has a charge of +3.
When aluminum atom loses its valence electrons, the charge on the resulting ion is 3+ .
Is aluminum negatively charged?
It is also important to note that the aluminum can is not a charged object. While it contains charges, it does not have an overall charge. The number of positive and negative charges is equal. This balance of the two types of charges is evidence that the aluminum can is neutral.
Is aluminum an atom or ion?
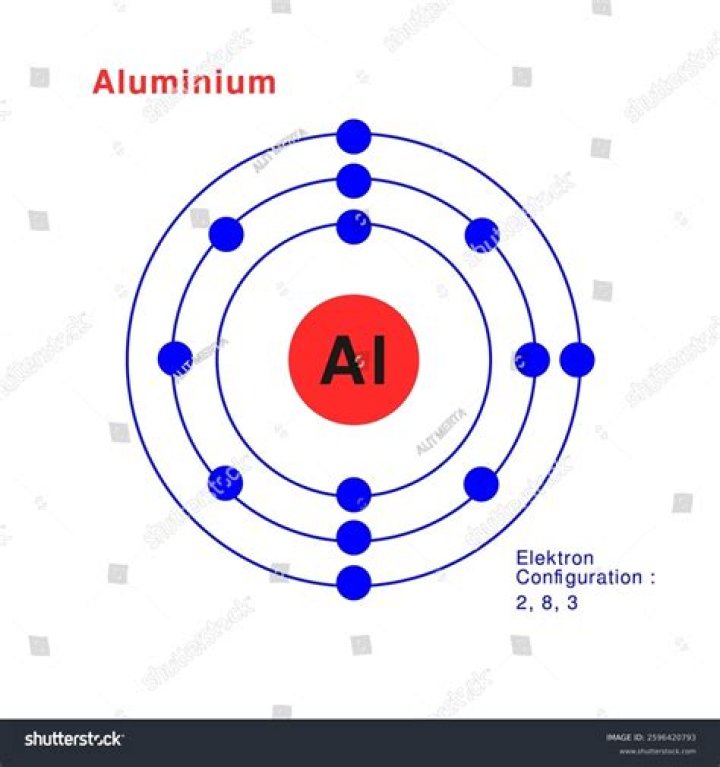
Given the fact that every proton has a positive charge, and that most atoms tend to be neutral in charge, this means that there are usually 13 electrons, with a negative charge, present in an atom of aluminum. Yet like all metals, aluminum is capable of forming an ion by losing electrons—in this case, three.
Once one shell is full, the next electron that is added has to move to the next shell. So for the element of ALUMINUM, you already know that the atomic number tells you the number of electrons. That means there are 13 electrons in a aluminum atom.
What’s a positively charged ion?
A cation is a positively charged ion with fewer electrons than protons while an anion is negatively charged with more electrons than protons. Because of their opposite electric charges, cations and anions attract each other and readily form ionic compounds.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
