What is the difference between NO2 and NO2 −?
The key difference between nitrite and nitrogen dioxide is that the nitrite is an anion whereas the nitrogen dioxide is a molecule. Both nitrite and nitrogen dioxide has the same number of nitrogen and oxygen atoms; one nitrogen atom and two oxygen atoms.
What is the name of NO2 )-?
Nitrogen Dioxide (NO2) is one of a group of highly reactive gases known as oxides of nitrogen or nitrogen oxides (NOx). Other nitrogen oxides include nitrous acid and nitric acid.
What is an NO2 minus?
Nitrite | NO2- – PubChem.
There are a total of 18 valence electrons for the Lewis structure for NO2-. Also note that you should put the NO2- Lewis structure in brackets with as 1- on the outside to show that it is an ion with a negative one charge.
Is NO2 negative polar?
NO2 (or nitrogen dioxide) is neutral as a gas, but can easily become NO2- (nitrite ion) in solution, demonstrating its polar nature. It is polar. Answer: NO2 is polar molecule due to the presence of a lone pair of electrons whose electron-electron repulsion results in a bent structure.
What Is Nitrogen Dioxide? Nitrogen dioxide, or NO2, is a gaseous air pollutant composed of nitrogen and oxygen and is one of a group of related gases called nitrogen oxides, or NOx. NO2 forms when fossil fuels such as coal, oil, gas or diesel are burned at high temperatures.
Is NH3 charged?
The formal chemical charge of Ammonia (NH3) is zero, it doesn’t actually have a chemical charge.
Why does NO2 have a negative charge?
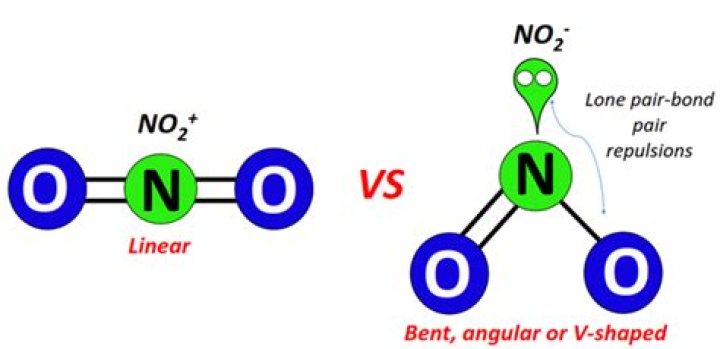
In nitrite ion, one oxygen atom is bonded to nitrogen by a double bond, while the other is bonded by a single bond. The oxygen atom bonded by a single bond has only one electron involved in bonding, and so contains one unpaired electron. As a result of this, the nitrite ion has an overall charge of -1.
Nitrous oxide is a safe and effective sedative agent that is mixed with oxygen and inhaled through a small mask that fits over your nose to help you relax. Nitrous oxide, sometimes called “laughing gas,” is one option your dentist may offer to help make you more comfortable during certain procedures.
Why is NO2 neutral?
To give a acidic solution the compound should have an ability to release hydronium ion. For that, water should be hydrolyzed and should form hydronium ion. But polarization of atoms of nitrogen dioxide and carbon monoxide is not enough to hydrolyze water. So they are neutral oxides.
How many pi bonds does NO2 have?
Hybridization of NO2
The single bond in N-O has one sigma bond and the double bond N=O. has one sigma bond and one pi bond.
Why does nitrite have a 1 charge?
The oxygens which have double-bonds have a shared or owned eight electrons, so they are considered neutral. Finally, the single bound oxygen atoms have nine electrons linked with them, and they have a negative charge overall. This means the nitrate ion has an overall charge of -1.
In NO2+ ion, the electronegativity of oxygen is 3.44 and that of Nitrogen is 3.04. Due to this difference, the NO bond ensures some non-zero dipole moment in the direction of Oxygen.
What is the electronegativity difference of n20?
The central N is bonded to different elements, N and O, making it polar. Furthermore, the N–N triple bond has no dipole moment (ΔEN = 3.04 – 3.04 = 0.0) while the N–O bond has one (ΔEN = 3.44 – 3.04 = 0.40). This results in a net dipole in the molecule.
What is the electronegativity difference of nitrogen oxide?
Electronegativity of oxygen(3.5) is higher than nitrogen(3.0).
Why is NO2 paramagnetic?
Answer: In gaseous state, NO2 exists as a monomer which has one unpaired electron but in solid state, it dimerises to so no unpaired electron left. Therefore, NO2 is paramagnetic in gaseous state but diamagnetic in solid state.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
