What is the electronic configuration of Cr minus?
The electronic configuration of Cr is (atomic number = 24): (a)- 1s22s22p63s23p64s24p4.
How do you write the electron configuration for Cr2+?
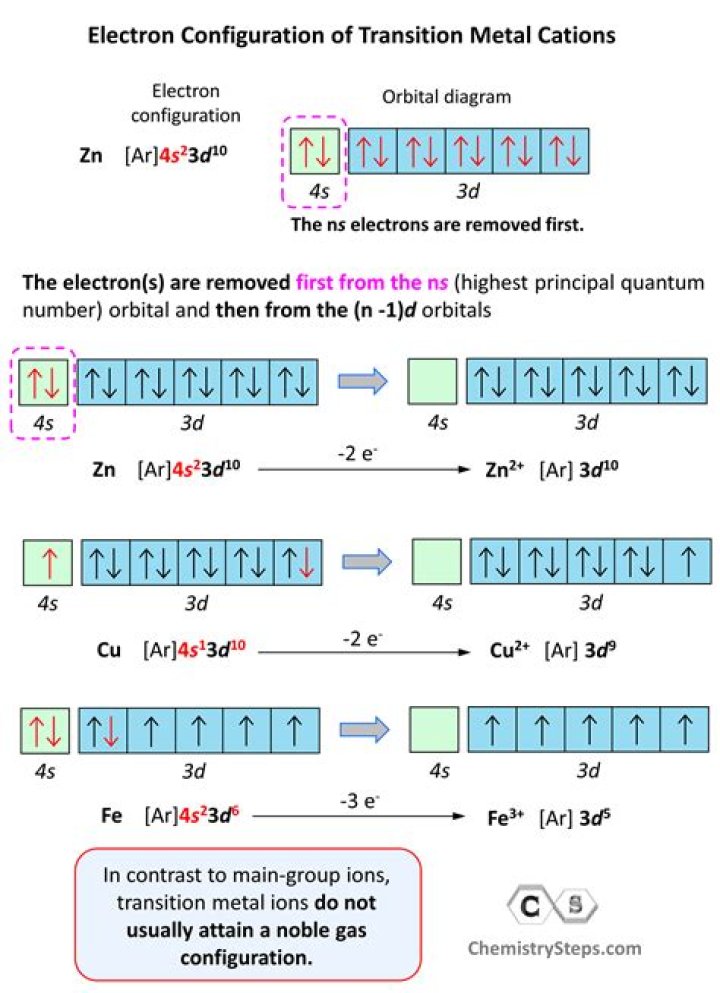
Answer: The electron configuration for Cr is [Ar]3d6 4s2 (note that 4s is higher in energy than 3d – that is important). Cr2+ means we remove 2 electrons, and we remove them from the highest energy level, which is 4s, so the electron configuration for Cr2+ is [Ar]3d6.
What is electronic configuration of Cr3+?
What is the electron configuration of Cr 3+? The electronic configuration of Cr having atomic number of 24 is 1s22s22p63s23p64s13d5 which is half-filled d-orbital. Cr3+ has 3 electrons removed from the outermost shell. Therefore, the electronic configuration comes out to be [Ar]3d3.
Cr3+ is larger than Cr and Se is larger than Se2-.
Which of the following are correct electron configurations for Cr?
So, the configuration of chromium is $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}4{{s}^{1}}$. Hence, the correct answer is an option (c)- $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}4{{s}^{1}}$.
chromium (Cr), chemical element of Group 6 (VIb) of the periodic table, a hard steel-gray metal that takes a high polish and is used in alloys to increase strength and corrosion resistance.
What does the electronic configuration 1s2 2s2 2p5 3s1 indicate?
Ground state of flurine atom. The ground state of neon is 1s22s22p6.
How do you write electronic configuration?
While writing electron configurations, a standardized notation is followed in which the energy level and the type of orbital are written first, followed by the number of electrons present in the orbital written in superscript. For example, the electronic configuration of carbon (atomic number: 6) is 1s22s22p2.
There are two main exceptions to electron configuration: chromium and copper. In these cases, a completely full or half full d sub-level is more stable than a partially filled d sub-level, so an electron from the 4s orbital is excited and rises to a 3d orbital.
How many 3d electrons are in Cr?
An atom Cr has one 4s electron and five 3d electrons.
What is the electronic configuration of Cu 2?
The electronic configuration of Cu2+ is [Ar]3d9.
How can we calculate Valency of CR?
Answer: Atomic number of chromium is 24. Chromium has six valence electrons in its outer most orbit. Hence its valency is 2.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
