What is the formula for lead IV oxide?
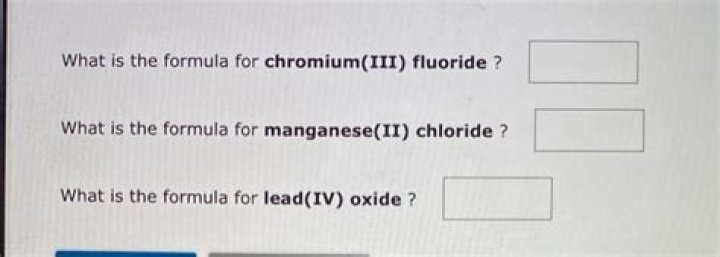
Lead IV Oxide features two compounds Pb4+ and O2− in the ratio 1:2 and hence the formula is PbO2.
What is the formula for lead IV nitride?
Lead(IV) Nitride Pb3N4 Molecular Weight — EndMemo.
How do you name PbSO4?
Lead sulfate | PbSO4 – PubChem.
Is PbSO4 a covalent compound?
In PbzO 4, oxygen forms covalent bonds with two Pb Il atoms if it is an apical oxygen, and with one Pb II if it is an equatorial oxygen.
What is the correct name of NH4 3PO4?
Ammonium phosphate is the salt of ammonia and phosphoric acid. It has the formula (NH4)3PO4 and consists of ammonium cations and phosphate anion.
What is the name of Pb OH Cl?
Lead oxychlorides are halide compounds of lead with the general formula PbClO(H).
What is the name of HgO?
Mercury(II) oxide, HgO, provides elemental mercury for the preparation of various organic mercury compounds and certain inorganic mercury salts.
What is FeO2 chemistry?
Iron dioxide | FeO2 – PubChem.
What is the correct name for V3 PO4 5?
Vanadium(V) Phosphate V3(PO4)5 Molecular Weight — EndMemo.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
