what is the formula for specific heat, check these out | What is the formula for calculating specific heat?
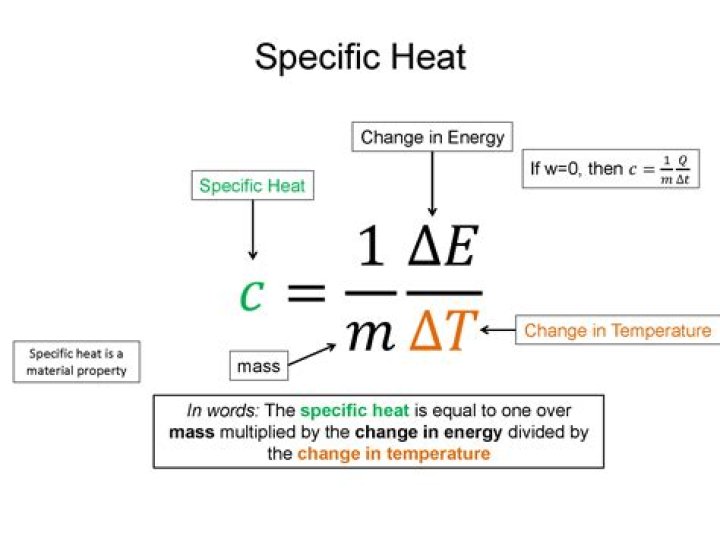
What is the formula for calculating specific heat?
The specific heat capacity is the heat or energy required to change one unit mass of a substance of a constant volume by 1 °C. The formula is Cv = Q / (ΔT ⨉ m) .
What is Q MCP ∆ T?
Q = mCpdT shows the heat required to raise the temperature of a material through a finite temperature difference (dT). Q = UA (LMTD) shows the rate of heat transfer across a finite temperature difference between 2 surfaces or fluids for a given surface area.
What is specific formula?
In simple words, the specific heat equation or the specific heat formula is the ratio of the amount of heat required to raise the temperature of a substance by one degree celsius to the amount of heat required to raise the temperature of the equal amount of water by one-degree Celsius at the room temperature.
How do you find q in specific heat?
Q = m•C•ΔT
where Q is the quantity of heat transferred to or from the object, m is the mass of the object, C is the specific heat capacity of the material the object is composed of, and ΔT is the resulting temperature change of the object.
What are the 3 formulas of heat?
Different formulas for Heat
H = (VI)t.H = (I 2 R)t.H = (V 2 /R)t.
What is the SHC of water?
The exact value of the specific heat capacity of water is 4182 J/kg°C. Now, water is quite commonly occurring and an important substance in our life. Therefore, there is a special way to identify the total amount of heat energy needed to raise one gram of water by a calorie (one degree Celsius).
What is specific heat with example?
specific heat, the quantity of heat required to raise the temperature of one gram of a substance by one Celsius degree. The units of specific heat are usually calories or joules per gram per Celsius degree. For example, the specific heat of water is 1 calorie (or 4.186 joules) per gram per Celsius degree.
How do you calculate Q reaction?
Calculate Q for a Reaction
Is given by: So essentially it’s the products multiplied together divided by the reactants multiplied together, each raised to a power equal to their stoichiometric constants (i.e. the numbers of each component in the reaction).
What does Q MCAT stand for?
Q = Heat energy (in Joules, J) m = Mass of a substance (kg) c = Specific heat (J/kg∙K) ∆T = Change in temperature (Kelvins, K)
What is the formula of heat and temperature?
The quantitative relationship between heat transfer and temperature change contains all three factors: Q = mcΔT, where Q is the symbol for heat transfer, m is the mass of the substance, and ΔT is the change in temperature.
What is specific heat of air?
The nominal values used for air at 300 K are CP = 1.00 kJ/kg. K, Cv = 0.718 kJ/kg.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
