What is the formula for zinc hydrochloric acid?
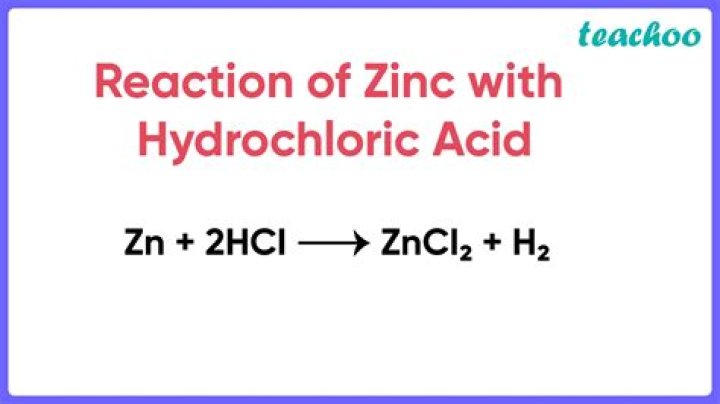
The reaction between zinc and HCl is given by the symbolic equation Zn + HCl → ZnCl2 + H2.
What happens when Zn reacts with HCl?
When zinc reacts with hydrochloric acid it produces zinc chloride and hydrogen gas.
Does Zn and HCl form a precipitate?
When zinc reacts with hydrochloric acid, the reaction bubbles vigorously as hydrogen gas is produced. A precipitate is a solid product that forms from a reaction and settles out of a liquid solution.
What is the net ionic?
The net ionic equation is the chemical equation that shows only those elements, compounds, and ions that are directly involved in the chemical reaction. Balancing by charge means making sure that the overall charge is the same on both sides of the equation.
Can there be no net ionic equation?
If you don’t have a net ionic equation to balance, that means you have what is usually called a molecular equation (or a complete molecular equation). These are harder to balance because the elements being oxidized and reduced are mixed in with substances that are are not being reduced or oxidized.
What type of reaction is zinc hydrochloric acid?
This is a double replacement reaction, that occurs spontaneously (exothermic reaction). Zinc reacts with hydrochloric acid to produce hydrogen gas and aqueous zinc chloride.
Why does zinc corrode in hydrochloric acid?
Since zinc is readily soluble in aqueous acidic solutions with the liberation of hydrogen the rate of hydrogen liberation corresponds to zinc corrosion rate.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
