What is the hybridization of CH4?
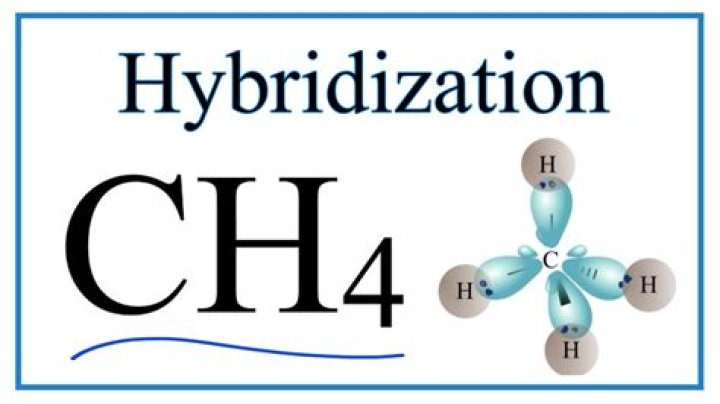
The type of hybridization involved with CH4 is sp3.
What is the the shape molecular geometry of NH4+?
According to the VSEPR theory, the NH4+ molecule ion possesses tetrahedral molecular geometry. Because the center atom, nitrogen, has four N-H bonds with the hydrogen atoms surrounding it. The H-N-H bond angle is 109.5 degrees in the tetrahedral molecular geometry.
What are sp2 hybridized carbons?
Hybridization – Carbon
A carbon atom bound to three atoms (two single bonds, one double bond) is sp2 hybridized and forms a flat trigonal or triangular arrangement with 120° angles between bonds.
What is hybridization of NH3?
In Ammonia (NH3) or to be more precise the central atom in ammonia which is nitrogen is sp3 hybridized.
Is NH4+ symmetrical or asymmetrical?
The nitrogen atom in the NH4+ molecule is also stabilized as a result of this. All the four N-H bonds in NH4+ ion are arranged in a tetrahedral pattern. H-N-H has a bond angle of about 109.5 degrees which pure tetrahedral structure. Each of the four N-H bonds in NH4+ ion is symmetrical to the others.
Is NH4+ linear?
The geometry is tetrahedral and since it contains two lone pairs which do not contribute to shape , the shape is bent.
What is the molecular geometry of NH4+ quizlet?
Ammonium, NH4+, has a central nitrogen atom surrounded by four hydrogen atoms. It has a tetrahedral electron geometry and a tetrahedral molecular shape.
What is the hybridization of NO2?
In the classical sense, NO2 is sp2 hybridized. In general, the Single-electron orbitals are unhybridized, and pure p-orbitals, like in methyl free radicals. However, a Single-electron orbital is known to be hybridized, when the central atom is bonded to the highly electronegative groups or atoms.
How do you find the hybridization of no3?
The hybridization of NO3– is sp2 type.
Important Points To Remember
The central atom nitrogen is bonded with three oxygen atoms and there are no lone pairs present.Nitrogen’s three sp2 orbitals overlap with one s orbital of the oxygen atom.The p orbital of nitrogen forms a double bond with three oxygen atoms.
Is NH4+ nonpolar or polar?
As the shape of the NH4 is tetrahedral all the four bonds are equivalent to each other so NH4 is Polar covalent bond.
Is NH4 ionic or covalent?
You are correct in thinking that NH4+ is covalent. However, this only applies to the bonds that make up the NH4+ itself. The molecule as a whole is charged, making an ion and thus, will form ionic bonds to non-metals (such as Cl-, O2-, and F-).
What is the dipole moment of NH4?
Similarly, the dipole of NH4+ (Ammonium ion) is 0 D.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
