What is the hybridization of iodine in IF3 and IF5?
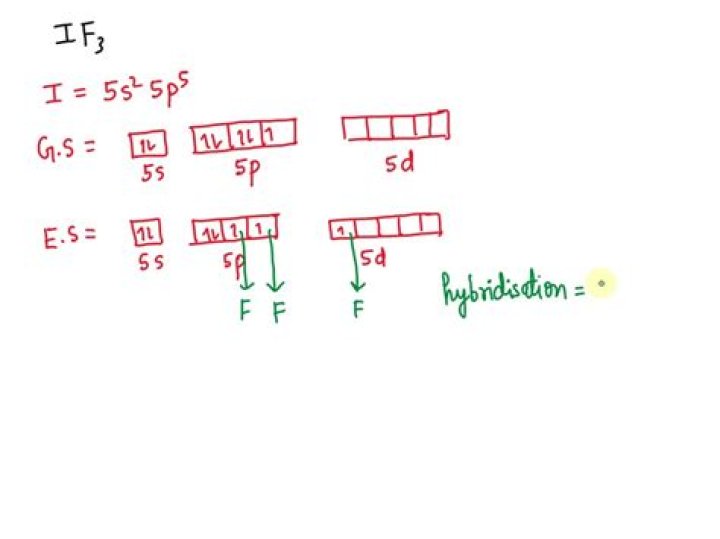
The hybridization of IF3 can be determined by the number of lone pairs around iodine and the number of sigma bonds formed between I and F. Since the iodine has 7 valence electrons out of which 3 electrons form sigma bond with F atoms and left with forming 2 lone pair.
What is the hybridization of iodine in IF3 and IF5?
Question: The hybridization of iodine in IF3 and IF5 are ___ and ____ respectively. The answer is sp3d,sp3d2I need a good explanationAlso this question as well:In the valence bond description of HCN, in which hybrid orbitals are the four electron pairs around N place if N is hybridized?
What is the hybridization of IF5?
H=6, the molecule will have Sp3d2 hybridization. Since the total charge of IF5 is 0, C and A will be zero. H=6, indicating that its Sp3d2 hybridized.
In which excited state iodine shows sp3d3 hybridization?
In third excited state , Iodine ( I ) shows sp3d3 hybridization . Seven half filled orbitals are generated in this configuration . This configuration results in sp3d3 hybridization . IF7 is a popular compound of Iodine in this hybridization .
Is IF3 polar or nonpolar?
IF3 is a polar molecule due to electronegativity difference Between I & F atom. The electronegativity values of I & F are 2.5 & 4.0 and the electronegativity difference betn I & F is (4.0–2.5) or 1.5.
How is hybridization calculated in IF3?
It is bonded to 3 atoms of F and also has 2 lone pairs on it. What is this? So when we add the number of bonded sites and lone pairs we get the value of H as 2+3 = 5, which means that IF3 is Sp3d hybridized.
What type of compound is IF3?
Iodine trifluoride is an interhalogen compound with the chemical formula IF3. It is a yellow solid which decomposes above −28 °C.
How many lone pairs are in IF3?
IF3 is dsp3 hybridized and contains 2 lone pairs and 3 bonding pairs of valence electrons around the Iodine. The VSEPR predicts the T-shape.
What is hybridization of iof5?
Since the central atom iodine has 7 valence electrons out of which 5 electrons form 5 sigma bonds with F atoms and and 2 electrons form 1 lone pair making the stearic number 6 which implies the hybridization of the central atom is sp3d2 where the geometry is octahedral and the shape is square pyramidal.
What shape is feco5?
In Fe(CO)5, the Fe-atom is in dsp3 hybridised state, therefore the shape of molecule is trigonal bipyramidal.
What is the hybridization of AsF5?
The hybridization of AsF5 is Sp3d as its central atom steric number is 5.
Is IF3 trigonal pyramidal?
According to ${text{VSEPR}}$, Theory, geometry of $I{F_3}$is trigonal bi pyramidal.
What is the name of IF3?
iodine(III) fluoride.
What is the hybridization of I3?
I3- ion is basically sp3d hybridized.
What type of hybridization is involved in SF4?
The hybridization that is involved in SF4 is sp3d type.
Which type of hybridization occurs in IF7?
The hybridization of central atom in IF7 is – sp3d3. IF7 has seven bond pairs and zero lone pairs of electrons.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
