What is the hybridization of SF4?
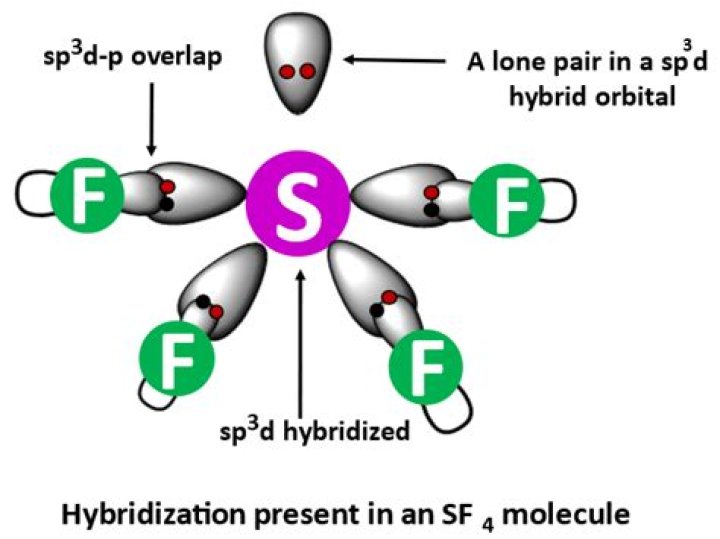
The hybridization that is involved in SF4 is sp3d type.
What is the hybridization of pf5?
The hybridization is sp3d hybridization and phosphorous atom forms five sp3d hybrid orbitals. Five hybrid orbitals will be used to form bonds with five fluorine atoms. There are 5 sigma bonds in this compound.
What is sp3d2 hybridisation?
Intermixing of one ‘s’, three ‘p’ and two ‘d’ orbitals of almost same energy by giving six identical and degenerate hybrid orbitals is called sp3d2 hybridization. These six sp3d2 orbitals are arranged in octahedral symmetry by making 90° angles to each other. Thus, SF6 molecule has octahedral structure.
How many sigma bonds does sf5 have?
Accroding to the Lewis structure of SF−5 S F 5 − ion is (given below), the central S atom is forming five sigma bonds with the five fluroine
What is the hybridization of I3?
I3- ion is basically sp3d hybridized.
Is PF5 sp3d2 hybridization?
– sp3d2 hybridization refers to 6 sets of electron pairs depicting octahedral structure. – Therefore, PF5 doesnt display sp3d2 hybridization.
Does PF5 have sp2 hybridization?
there are 5 sigma bonds in this compound. hence , the hybridization of this compound is sp3d . 3 has sp2 hybridization.
What is the hybridization of C in ch4?
Now coming to the hybridization of methane, the central atom carbon is sp3 hybridized. This is because one 2s orbital and three 2p orbitals in the valence shell of carbon combine to form four sp3 hybrid orbitals which are of equal energy and shape.
How many Unhybridized orbitals are in sp3d?
– sp3 hybridization has 4 sp3 hybridized orbitals and 0 unhybridized orbitals. These allow molecules to form 4 sigma bonds.
What will be the hybridisation of Xe in XeF2?
Hence the hybridization of the central atom Xe is sp3d. Thus the hybridization of XeF2 molecule is sp3d.
In which of the following Xe atom is sp3d2 hybridization?
Xenon will require its s orbital along with its p-orbitals which are three in number, and 2 of its d-orbitals to form the hybridization state as sp3d2, or d2sp3.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
