What is the molecular geometry of CH3Br?
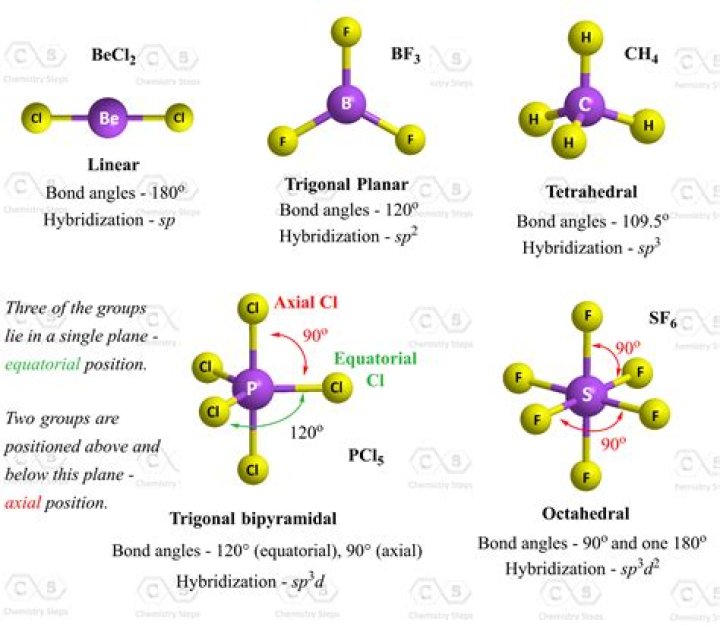
According to the VSEPR theory, CH3Br possesses tetrahedral molecular geometry and CH4-like electron geometry. Because the center atom, carbon, has three C-H and C-Br bonds with the hydrogen and bromine atoms surrounding it. The H-C-Br bond generates 111.5 degrees in the tetrahedral molecular geometry.
Is CH3Br tetrahedral?
The CH3Br molecule is polar. Both CBr4 and CH3Br have four regions of electrons around the central carbon atom. These are all bonding electron regions (clouds) so the shape of both molecules is tetrahedral.
How many valence electrons does CH3Br?
For CH3Br, there are a total of 14 valence electrons. Hydrogen always goes on the outside, and since Carbon is less electronegative than Bromine, we’ll put the Carbon in the center and the Bromine on top. We have 14 valence electrons for the CH3Br Lewis structure. Put two between atoms to form chemical bonds.
How many bonds does CH3Br have?
Chemical Structure Description
The Bromomethane molecule contains a total of 4 bond(s) There are 1 non-H bond(s).
Is CH3Br polar or non polar?
The CH3Br molecule is polar. Both CBr4 and CH3Br have four regions of electrons around the central carbon atom. These are all bonding electron regions (clouds) so the shape of both molecules is tetrahedral. The C-Br bond is polar due to the difference in electronegativity between C and Br.
How many double bonds does CHBr3 have?
The CHBr3 molecule has a tetrahedral molecular geometry because there is no electrical repulsion between the lone pairs of electrons in bromine and four bond pairs(C-H and three C-Br) of the CHBr3 molecule. Lewis structure of CHBr3 has dot electron representative structure.
What is the bond angle for a tetrahedral?
The proof that a bond angle in a tetrahedral molecule is 109.5 degrees is more complex than it first appears.
How many lone pairs of electrons are in the Lewis structure of CH3Br?
Methyl bromide (CH3Br or bromoethane) is an alkyl halide compound. It has only one carbon atom. Lewis structure of CH3Br contains 3 C-H bonds and 1 C-Br bond. Carbon atom is the center atom and bromine atom has 3 lone pairs.
Does CH3Br have dipole dipole forces?
CH3Br is a polar molecule. Dispersion forces (present in all matter) and dipole-dipole forces will be present. This compound has the next highest boiling point. The only intermolecular force present is a relatively weak dispersion force (smaller molar mass).
Is CH3Br organic?
Learn about the dangers facing strawberry crops and the measures growers are taking to protect them. methyl bromide, also called bromomethane, a colourless, nonflammable, highly toxic gas (readily liquefied) belonging to the family of organic halogen compounds.
Why does CH3Br have a higher boiling point than ch3i?
If we compare methyl bromide and methyl iodide , methyl iodide has less boiling point ,since Br is more electronegative, it withdraws more electron towards itself and hence increase dipole moment within the atom . Therefore . it requires more energy to split them up and that indicates higher boiling point.
What is the dipole moment of CH3Br?
The dipole moment of the isolated molecular adsorbates obtained from the best fit are 2.57 ( 0.05 and 2.12 ( 0.05 D for CH3Br and CH3Cl, respectively.
Is AsF3 a dipole?
AsF3 is polar because it has a lone pair, so the dipoles do not completely cancel. All the dipoles in AsF5 cancel, that’s why it is non polar. Because AsF3 is polar, it has dipole-dipole intermolecular forces, so its boiling point would be higher.
Which type of bond is present in the compound CH3Br?
Bromomethane is a one-carbon compound in which the carbon is attached by single bonds to three hydrogen atoms and one bromine atom.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
