What is the molecular geometry of teo2?
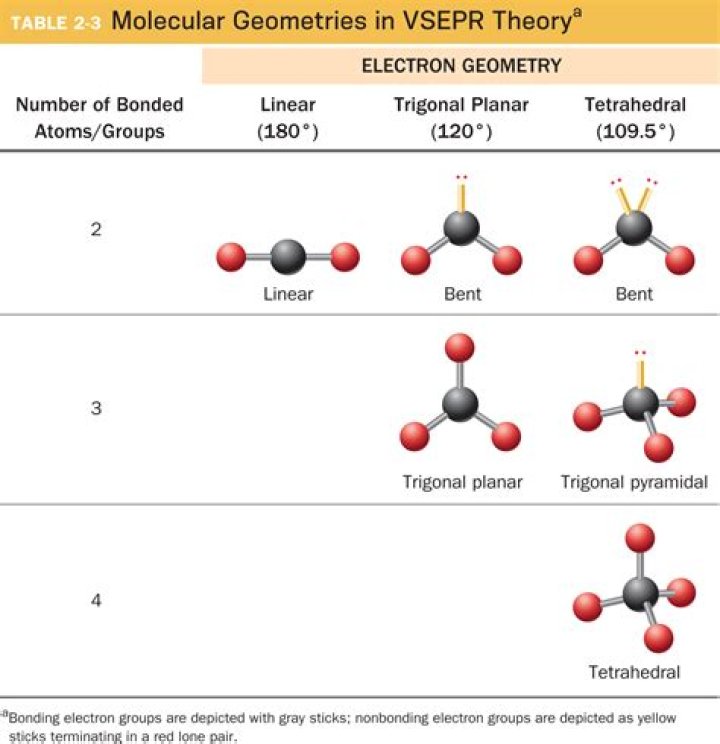
The molecular geometry of OF2 is bent and its electron geometry is tetrahedral because the presence of two lone pairs on the central atom creates repulsion with bonded pairs of electrons, as a result, all outer atoms(fluorine) pushes down in order to minimize the repulsion according to the VSEPR theory, and that makes …
What is the molecular geometry of teo2?
This molecule is trigonal planar because it has 3 e- pairs around the central atom, 3 atoms bonded to it, and 0 lone pairs.
What is the molecular geometry of a molecule with 2 outer atoms and 1 lone pair on the central atom?
Nathan L. This is a bent molecule.
What kind of bond is in OF2?
In OF2 O F 2 , two O-F covalent bonds are present. The electronegativity of oxygen is 3.5. The electronegativity of fluorine is 4.0.
Is TeO2 linear?
Tellurium dioxide TeO2 in crystalline and glassy state attracts considerable attention as a material with outstanding non-linear optical (NLO) properties. The third-order non-linear refractive index of glassy TeO2 is 50 times higher than that of the silica glass [1].
Is TeO2 polar?
Tellurium oxide is probably polar covalent; tellurium is of moderate electronegativity and oxygen is of high electronegativity.
Why is the molecular geometry of a water molecule bent?
Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). The molecule adopts a bent structure because of the two lone pairs of electrons on the oxygen atom.
Why does OF2 not have a double bond?
If you examine the structure of OF2, both are second period elements so these elements have “no” d- orbitals (their valence orbitals are only one 2s orbital and three 2p -orbitals.) Each fluorine here, forms one sigma bond and has three lone pairs.
What is the geometry of the SF6 molecule its Lewis structure is?
The molecular geometry of SF6 is octahedral with a symmetric charge distribution around the central atom.
Is TeO2 a solid?
Tellurium Dioxide, has the symbol TeO2 is a solid oxide of tellurium.
Is NH2Cl polar or nonpolar?
NH2Cl is a polar molecule because of its asymmetrical shape i.e. trigonal pyramidal that ensures that vectors will not cancel out in this molecule which results in some net dipole moment that makes NH2Cl polar in nature. Basically, NH2Cl forms three types of bonds – two N-H and one N-Cl.
Why TeO2 is an oxidising agent?
electrons are reluctant to take part in bond formation due to poor shielding of d- and f-orbitals thus lower oxidation states (+2, -2) becomes more stable( inert pair effect) and hence TeO2 is oxidising agent.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
