What is valency of oxygen?
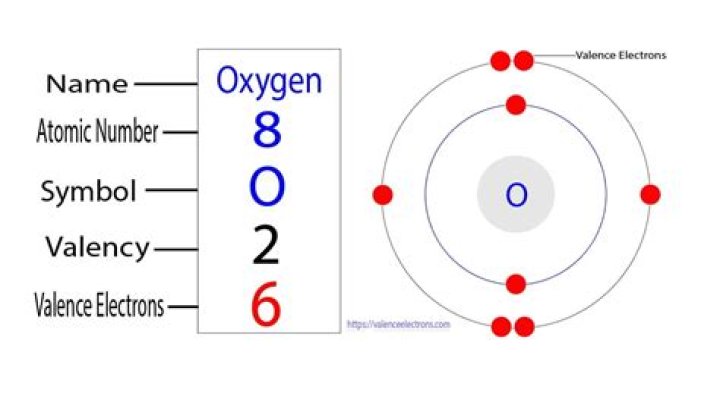
The valency of oxygen is 2, because it needs two atoms of hydrogen to form water. … The nearest noble gas to magnesium is neon with electronic configuration of [2,8], to achieve this stable electronic configuration Mg can lose 2 valence electrons, hence its valency is 2 + ^+ + .
What is the valency of oxygen Brainly?
The valency of oxygen is 2 as its electronic configuration is 2,6 and it need 2 electron to complete their octet.
What is meant by valency?
Valency is the combining power of an element. Elements in the same group of the periodic table have the same valency. The valency of an element is related to how many electrons are in the outer shell. Group number.
From the electronic configuration of the oxygen atom, we come to know that the number of electrons present in the outermost shell i.e., 2rd shell is 6 electrons. Thus, the total number of valence electrons present in an oxygen atom is six.
How can we find valency?
Mathematically we can say that if the outermost shell of an atom contains 4 or less than 4 electrons, then the valency of an element is equal to the number of electrons present in the outermost shell and if it is greater than 4, then the valency of an element is determined by subtracting the total number of electrons
The hydrogen atoms are each sharing one pair of electrons – they have a valency of 1. The oxygen atom is sharing two pairs of electrons – it has a valency of 2.
What is the valency of oxygen atomic no of oxygen 8?
Atomic Number of oxygen is 8. So, electronic Configuration of oxygen= 2, 6. So.. valency is 8-6 = 2.
What is carbon valency?
Now, we know that the number of electrons in the outermost shell of carbon is 4. In order to achieve the noble gas configuration of the nearest noble gas, carbon makes four bonds with hydrogen. Therefore, the valency of carbon is 4.
The charge of Aluminium is +3 and the charge of Oxygen is -2. Therefore, we can conclude that the valency of Aluminium in Al2O3 is 3 and the valency of Oxygen in Al2O3 is 2.
Does oxygen have variable valency?
Certain elements have more than one valency. They exhibit variable valency. For example, copper, tin, iron and mercury exhibit variable valency. Valency of oxygen being two, combining capacity of copper in the first case is one and that in the second case is two.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
