What kind of bond is bf3?
It follows, therefore, that an atom will tend to make as many covalent bonds as possible. In the case of boron in BF3, three bonds is the maximum possible because boron only has 3 electrons to share. You might perhaps wonder why boron doesn’t form ionic bonds with fluorine instead.
Is BF3 polar or nonpolar covalent bond?
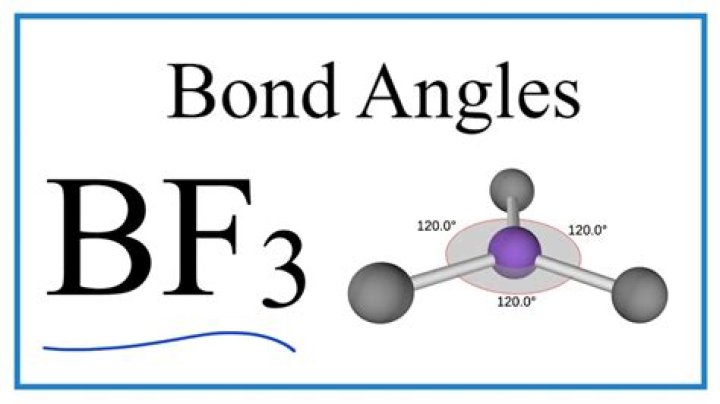
BF3 (Boron Trifluoride) is Non-Polar because of its highly symmetric shape. It has a Trigonal Planar geometry which cancels out the dipole moments of the three BF bonds making the resultant Dipole Moment of the compound equal to 0 (Zero).
Is BF3 covalent compound?
The molecule BF3 and NF3 , both are covalent compounds but BF3 is non – polar and NF3 is polar.
BF3 is an electron-deficient compound. Boron(B) atom has 3 electrons in its outermost shell and forms 3 single covalent bonds with fluorine and attains 6 electrons.
Is BF3 a dative bond?
One of the most well-known examples of dative bonding is the bond between ammonia (NH3) and boron trifluoride (BF3). The boron of BF3 has only six valence electrons and requires two more electrons to satisfy the octet rule.
Boron trifluoride (BF3) is a nonpolar molecule, whereas ammonia (NH3) is a polar molecule. The difference in polarity is related to the fact that: A. BF3.
Is BH3 polar or nonpolar?
BH3 (Borane) is a nonpolar substance because of the symmetrical trigonal planar geometry. The electronegativity of Boron(2.04) and Hydrogen(2.20) atoms is nearly the same due to which the B-H bond is almost nonpolar.
Does BF3 have ionic and covalent bonds?
BF3 is a molecule consisting of an sp2 hybrid of Boron covalently bonded with 3 atoms of fluorine. The covalent bond tells us that electrons are shared, rather than lost by boron and gained by fluorine. The simplest answer is that both atoms are small, therefore they make very strong covalent bonds.
Boron and chlorine are both nonmetals, so this is a covalent compound.
Is baf2 ionic or covalent?
Barium fluoride is an ionic compound because it is made up of the barium cation (has a +2 charge) and
What is the covalent compound name for BF3?
Boron trifluoride | BF3 – PubChem.
Is mgcl2 ionic or covalent?
As we can see that there is loss and gain of electrons between chlorine and magnesium so magnesium chloride is an ionic compound and there is electrostatic attraction between them so it is an electrovalent compound also.
Boron trifluoride BF3
But BF3 reacts very energetically with molecules such as water and ammonia that have available electron pairs (lone pairs). This indicates that BF3 is electron-deficient.
How is the bond between BF3 and F formed?
BF3 is a covalent molecule that reacts with an F- ion to form a BF4 – ion.
What are the types of covalent bond?
Types of Covalent Bonds
Single Covalent Bond.Double Covalent Bond.Triple Covalent Bond.
Is o3 coordinate bond?
Ozone does not have two coordinate bonds emanating from the central oxygen atom. Then only one of the oxygen atoms donate an extra electronic pair to a nearby oxygen radical (O°) to form ozone gas.
Because of the relatively high difference of electronegativity between the bromine atom and fluorine atoms, the bonds of the BrF3 are considered polar. Because of the presence of the two lone pairs in hybridization, the molecule’s shape is bent on certain bond angles.
What intermolecular forces are present in BF3?
As a result, the only type of intermolecular forces in BF3 would be the London dispersion forces.
Why BCl3 and BF3 are non-polar?
Boron Trichloride or BCl3 is a nonpolar compound because of its symmetrical structure ie; Trigonal Planar. The B-Cl bond itself is polar because of the difference in electronegativity of Boron(2.04) and Chlorine(3.16) atoms and all three B-Cl bonds lie at 120 degrees to each other.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
