What kind of bond is COCl2?
The two Chlorine atoms flank the Cobalt atom on either side. We then use the valence electrons to form covalent bonds between the atoms. Two valence electrons are used to create a Co-Cl bond.
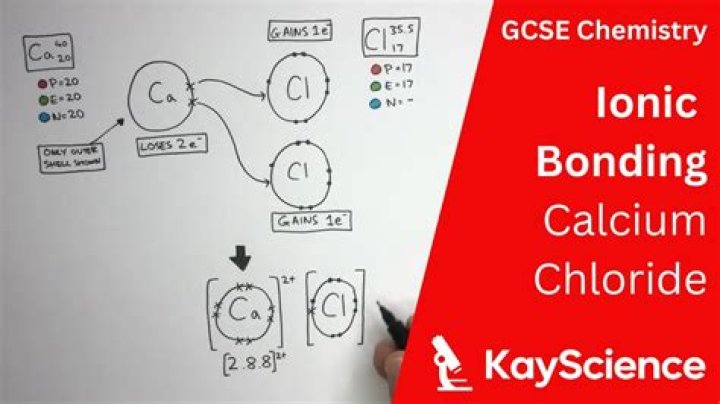
Is iron chloride an ionic or covalent bond?
Explanation: Iron (III) Chloride is an ionic compound, its formula unit is FeCl3 . This indicates that FeCl3 is the smallest repeating unit within the repeating crystal lattice structure of the compound. In general, ionic compounds are classified as ones that are composed of both a metal and a nonmetal.
Is CoCl2 a covalent bond?
CoCl2 Hybridization
Lone pairs and covalent bonds with other atoms contribute to being electron domains. Two domains give us an sp hybridization. Three domains give us and sp2 hybridization and so on. Therefore, in this case, there are two covalent bonds present, i.e., the two Co-Cl bonds.
Cobalt(II) chloride is an inorganic compound of cobalt and chlorine, with the formula CoCl2. It is a red crystalline solid. Cobalt chloride has been classified as a substance of very high concern by the European Chemicals Agency as it is a suspected carcinogen.
Is iron II chloride a covalent bond?
FeCl3 is an ionic compound, because iron (a metal) has bonded with chloride ions (non-metal element).
Covalent Bonding. Ionic compounds, such as sodium chloride (NaCl), are formed by a transfer of electrons that creates ions. The hydrogen and oxygen atoms in a water molecule, however, are bonded by sharing electrons rather than by transferring them.
Is iron covalent bond?
Iron has a type of bonding called metallic bonding. Iron is a metal and all metals have a type of bonding called metallic bonding which is different from covalent or ionic bonding. In metallic bonding the outer shells of adjacent atoms overlap, and the outer shell electrons are free to move through the lattice.
What is the molecular geometry of PH3?
The molecular geometry of PH3 is trigonal pyramidal. We start with the Lewis structure of PH3. The molecule has eight valence electrons. Phosphorus
More specifically speaking, carbon tetrachloride is a nonpolar covalent compound because the electrons shared by the carbon and chlorine atoms are nearly at the center of the bond. Therefore, the carbon tetrachloride ($CC{{l}_{4}}$) is a covalent compound.
Does fe2o3 have covalent bonds?
Iron oxide or ferric oxide has three oxygen atoms and two iron atoms. It is represented by Fe2O3. Since iron is metal and oxygen is non-metal therefore the bonding between oxygen and iron is ionic.
Is SO2 a covalent compound?
Sulfur dioxide is a Covalent compound because, in the case of sulfur dioxide, two atoms possessing similar electronegativity try to bond together. In doing so, the slight difference of electronegativity between the two atoms results in sharing of electron bonds forming covalent bonds.
What type of chemical bond is fe2o3?
Iron(III) oxide structure – Fe2O
The oxidation state of Fe2O3 is +3. The bond formation between oxygen and iron depends on the difference in electronegativity between these two atoms. Iron(Fe) is metal whereas oxygen(O2) is non-metal. Therefore, such bonds are called an Ionic bond.
Phosphine is the best example of a polar molecule having non-polar bonds. As with three hydrogen bonds and a lone pair, hydrogen and phosphorus are equal in electronegativity values.
Is CH4 a covalent or ionic compound?
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). The lines, or sticks, as we say, represent the covalent bonds. There are four bonds from a central carbon (C) linking or bonding it to four hydrogen atoms (H).
Is Li and O ionic or covalent?
Lithium oxide is an ionic compound formed between a metal (Li) and a non- metal (O) by the complete transfer of electrons from Li to O to give Li+ cations and O2– anions.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
