What kind of reaction is SO2 O2 SO3?
It is a combustion reaction, but also it is a redox reaction.
Is SO2 O2 endothermic or exothermic?
The oxidation of SO2 by O2 to SO3 is an exothermic reaction.
Is SO2 O2 a redox reaction?
The 105 systems involve six redox reaction types: SO2/O2; SO2/NOx; SO2/H2O2; SO2/O3; NOx/O3; NOx/H2O2. Mn(+2) and Fe(+2) are the only important metal catalysts.
When sulfur dioxide is added to oxygen sulfur trioxide is produced?
When sulfur dioxide is added to oxygen, sulfur trioxide is produced. Sulfur dioxide and oxygen, SO2+O2, are reactants and sulfur trioxide, SO3, is the product.
What is 2SO2 O2 → 2SO3?
2SO2 + O2 → 2SO3. Sulphur dioxide + oxygen → Sulphur trioxide.
Is SO2 combination reaction?
Sulfur reacts with oxygen to form sulfur dioxide. When nonmetals react with one another, the product is a molecular compound. Often, the nonmetal reactants can combine in different ratios and produce different products. Sulfur can also combine with oxygen to produce sulfur trioxide.
What type of reaction is SO2?
The creation of sulfur dioxide is an exothermic reaction (delta H -296), meaning that it gives off heat.
What happens to the equilibrium yield of Sulphur trioxide SO3 if the temperature is increased explain your answer?
Rates: At a low temperature, the formation of sulphur trioxide is very slow. Increasing the temperature increases the rate at which equilibrium is reached. However, a high temperature means a lowering of the percentage yield of sulphur trioxide.
How is yield of SO3 Maximised?
According to Le Chatelier’s Principle, increasing the concentration of oxygen in the mixture causes the position of equilibrium to shift towards the right. The 1:1 mixture results in the best possible overall yield of sulfur trioxide.
Which catalyst is used in oxidation of so2 to SO3?
Vanadium-based catalysts have well known uses in several reactions such as oxidative dehydration [1,2], oxidative dehydrogenation [3] and in the notable oxidation of sulfur dioxide to sulfur trioxide, a step in the production of sulfuric acid.
What is being oxidized and what is being reduced SO2 O2 SO3?
Sulfur is oxidized, and oxygen gas is REDUCED
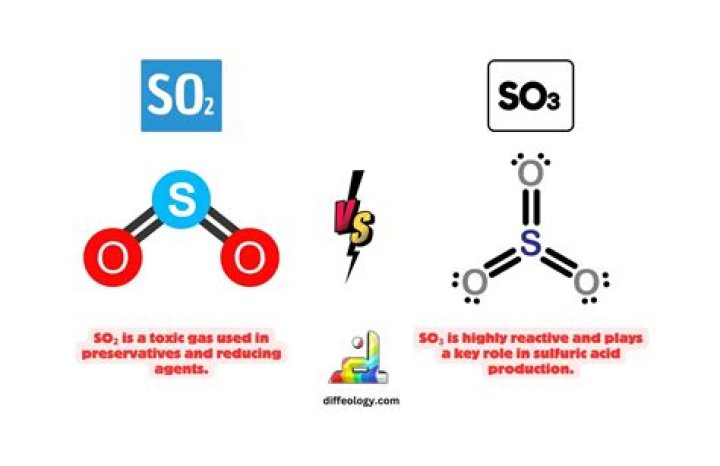
What is SO3?
Sulfur trioxide (SO3) is generally a colorless liquid. SO3 is also called sulfuric oxide and sulfuric anhydride. It is used in the production of sulfuric acid and other chemicals, and explosives. Sulfuric acid is a clear, colorless, oily liquid that is very corrosive.
What happens when SO2 reacts with oxygen?
Sulphur dioxide combines with oxygen to form sulphur trioxide.
When sulfur dioxide is added to oxygen sulfur trioxide is produced what is the correct chemical equation for this reaction?
The balanced equation is 2SO2+O2→2SO3 .
What happens when sulfur dioxide reacts with oxygen?
Formed by the reaction of sulfur dioxide and oxygen in the presence of catalysts, sulfur trioxide fumes vigorously in contact with moist air and dissolves in water, liberating much heat and forming sulfuric acid. Solutions of the trioxide in sulfuric acid are called fuming sulfuric acid, or oleum.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
