What leads to the formation of an ionic bond?
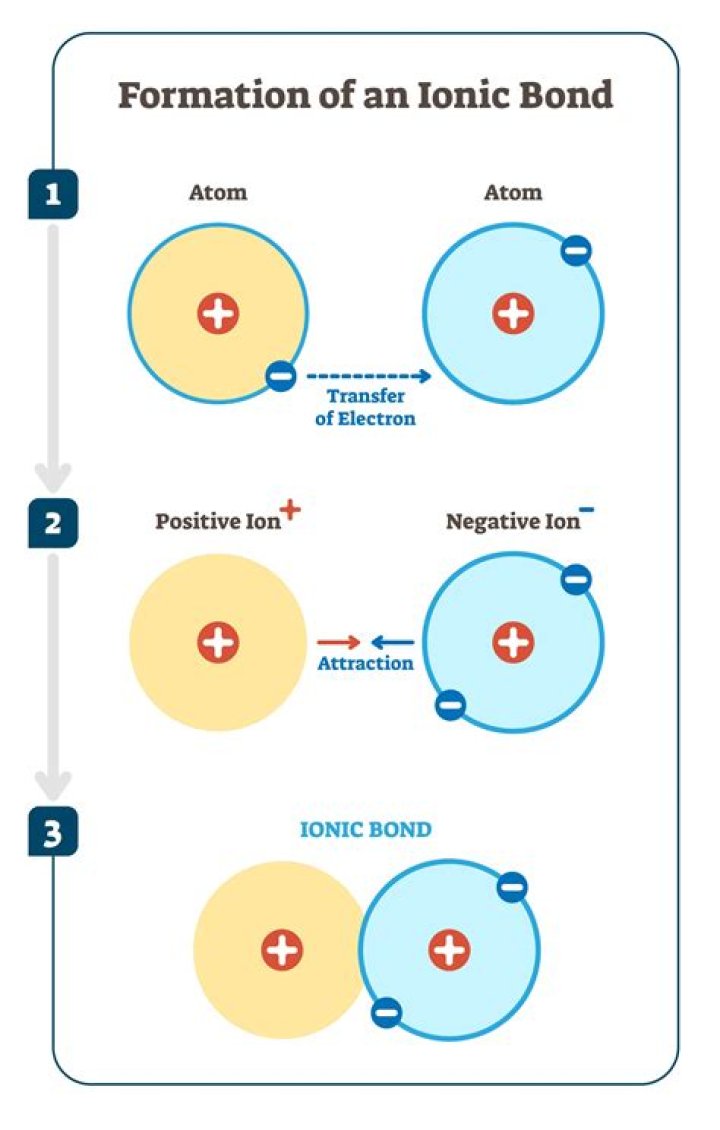
An ionic bond is formed by the complete transfer of some electrons from one atom to another. The atom losing one or more electrons becomes a cation—a positively charged ion. The atom gaining one or more electron becomes an anion—a negatively charged ion.
What leads to the formation of ionic bond with Hg2 plus?
What leads to the formation of an ionic bond with Hg2^2+? An attraction occurs between two ions that have the same charge. A metal atom forms a cation, and a nonmetal atom forms an anion. A more electronegative atom transfers electrons to a less electronegative atom.
Which pair of ions can form an ionic bond with each other?
For example, in the reaction of Na (sodium) and Cl (chlorine), each Cl atom takes one electron from a Na atom. Therefore each Na becomes a Na+ cation and each Cl atom becomes a Cl- anion. Due to their opposite charges, they attract each other to form an ionic lattice.
What is needed to form an ionic bond?
Ionic bonds require an electron donor, often a metal, and an electron acceptor, a nonmetal. Ionic bonding is observed because metals have few electrons in their outer-most orbitals. By losing those electrons, these metals can achieve noble gas configuration and satisfy the octet rule.
How are ionic bonds formed quizlet?
ionic bonds form when electrons are transferred from one atom to another atom. ions of different elements can combine by forming ionic bonds . positive ions & negative ions form when atom s lose or gain electrons.
How does an atom become a positive ion?
The atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called an anion). Opposite charges attract one another while similar charges repel one another.
How do ionic bonds affect the properties of ionic compounds?
How do ionic bonds affect the properties of ionic compounds? The bonds prevent electrons from moving throughout the crystal, so a solid ionic compound is a poor conductor. One compound is covalent, and the other is ionic.
How does lattice energy affect ionic bond formation?
The higher the lattice energy, the greater is the tendency of the formation of an ionic bond. The higher the charges on the ions and smaller the distance between them, the greater is the force of attraction between them.
What is formed when polyatomic ions bond with other ions?
-covalent bonding holds the atoms together within the ion and ionic bonding joins the entire ion to other oppositely charged ions. polyatomic ions can join with single atom ions or other polyatomic ions to form ionic compounds.
What holds ionic bonds together?
Oppositely charged particles attract each other. This attractive force is often referred to as an electrostatic force. An ionic bond is the electrostatic force that holds ions together in an ionic compound.
Which pair of ions can form an ionic bond with each other and why Cu and Ag They are both metal ions S and O they have like charges?
A metal atom forms a cation, and a nonmetal atom forms an anion. Which pair of ions can form an ionic bond with each other and why? Cu+ and Ag+; They are both metal ions. S 2- and O 2-; They have like charges.
When can ionic bonds be formed?
ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.
What are the factors governing the formation of an ionic bond explain the formation of an ionic bond with the help of an example?
Ionic bond formation mainly depends upon three factors –i Low ionization energy – elements with low ionization enthalpy have greater tendency to form an ionic bonds. ii High electron gain enthalpy – high negative value of electron gain enthalpy favours ionic bond.
Which molecule is formed by ionic bonds quizlet?
In an ionic bond, the atoms are bound together by the attraction between oppositely-charged ions. For example, sodium and chloride form an ionic bond, to make NaCl, or table salt.
Which option describes something that causes ionic bonds to form quizlet?
An ionic bond occurs when there is a large difference in electronegativity between two elements, such as often occurs between a metal and nonmetal, resulting in almost complete transfer of the electron from the metal to the nonmetal.
How is a cation formed?
A cation is an ion with positive charge, which means it has more protons (positively-charged particles) than electrons (negatively-charged particles). Cations are formed when an atom loses one or more electrons: the loss of the negatively-charged electron(s) results in an overall positive charge.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
