What type of bond is KCl?
KCl is highly polar as it is an ionic compound. The difference in electronegativity is very high and hence the percentage ionic character is high.
What type of bond is KCl?
Explanation: Potassium chloride, KCl , is an ionic compound formed by the electrostatic force of attraction that holds the potassium cations and the chlorine anions together.
Is the bond in potassium chloride KCl nonpolar polar or ionic?
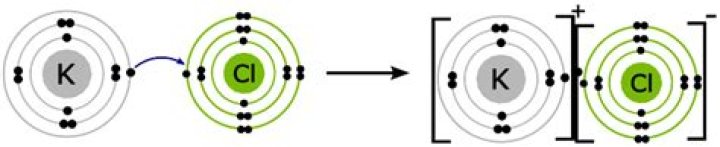
Potassium chloride is ionic. It consisted of potassium (K) and chlorin (Cl) atoms. K has 1 valence electron, while Cl has 7 valence electron. To fulfill Octet Rule, K will give one of its electrons to Cl which lacks one electron, forming an ionic bond.
Is KCl an ionic or covalent compound?
Its chemical formula is KCl, consists of one potassium (K) atom and one chlorine (Cl) atom. An ionic compound is made of a metal element and a nonmetal element. In potassium chloride, the metal element is potassium (K) and the nonmetal element is chlorine (Cl), so we can say that KCl is an ionic compound.
Is KCl stable compound?
The phase diagram shows that KCl remains stable in the whole pressure range investigated here, but many new compounds become stable at elevated pressures.
Why is KCl not a molecule?
It is incorrect because potassium and chlorine do not react to form molecules. There is no such thing as a “KCl molecule”.
Is CH3CH2OH polar or nonpolar?
The structure of ethanol is given below. Hence, the molecule CH3CH2OH C H 3 C H 2 O H is polar.
What is the electronegativity of KCl?
Calculate the percentage of ionic character in K-Cl bond in KCl molecule. The electronegativity values of K and Cl are 0.8 and 3.0 respectively.
Does KCl form a covalent bond?
Yes, KCl, or potassium chloride, is an ionic bond.
What type of chemical bond is ch3ch2oh?
The hydrogen-oxygen and carbon-oxygen bonds are polar covalent bonds.
Is KCl a metal or nonmetal?
Potassium chloride (KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine.
Why is KCl stable?
Because potassium have ability to stabilize itself along with positive charge more than sodium. so KCl is more stable than NaCl. One more reason is that the atomic radius of potassium is greater than that of sodium so it can donate or lose its electron easily and becomes stable with positive charge.
Is KCl soluble in ethanol?
As Chloride ion is smaller, the polarization is less and not much covalent character. Covalent substance disolve in organic solvents. Therefore, KI having some covalent character disolves but KCl doesn’t. Hope this helps!
Is KCl soluble in hexane?
Precipitation is a function of solubility
This is due to the fact the salts are not soluble in hexane and thus salt ions are not present in hexane.
Is CrCl3 ionic or covalent?
AlCl3, aluminum chloride; CrCl3, chromium(III) chloride; ICl3, iodine trichloride; AlCl3 and CrCl3 are ionic compounds following the rules for naming ionic compounds. The major difference is that CrCl3 contains a transition metal (Cr) which generally exhibits two or more stable charges when in ionic compounds.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
