What type of bond will sulfur S and fluorine F make?
No, fluorine and sulphur will create a covalent compound because ionic compounds are obtained from non-metal and metal whereas covalent compounds are
What is a Type 3 compound?
Binary Covalent Compounds (Type III)
These compounds are always neutral (not ions which have charges), and consist of only two elements (see acid naming below for compounds containing only non-metal elements, but with more than two elements. The prototypical compound is CO2, which is called carbon dioxide.
What is in sulfur dibromide?
Sulfur dibromide is the chemical compound with the formula SBr2. It is a toxic gas. Sulfur dibromide readily decomposes into S2Br2 and elemental bromine. In analogy to sulfur dichloride, it hydrolyzes in water to give hydrogen bromide, sulfur dioxide and elemental sulfur.
What is the hybridization of sf3?
hence, the hybridization of it is sp3d2. you can also see the hybridization process of IF5 in this picture.
What is sulphur fluoride?
Sulfuryl fluoride (also spelled sulphuryl fluoride) is an inorganic compound with the formula SO2F2. It is an easily condensed gas and has properties more similar to sulfur hexafluoride than sulfuryl chloride, being resistant to hydrolysis even up to 150 °C.
What is the name of Rb2O?
Rubidium oxide (Rb2O)
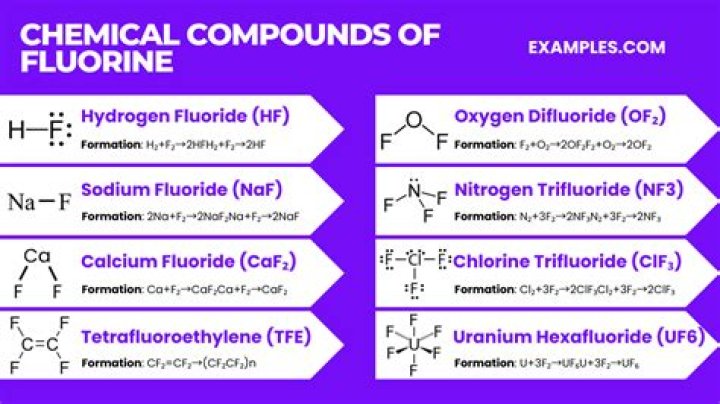
Is NaF a binary ionic compound?
We are provided with the binary compound NaF. It is made up of sodium metal ion and fluoride non metal ion.
What type of bonding is sulfur?
Each sulfur atom is bonded to each of its two neighbors in the ring by covalent S-S single bonds.
Is CH4 a covalent or ionic compound?
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). The lines, or sticks, as we say, represent the covalent bonds. There are four bonds from a central carbon (C) linking or bonding it to four hydrogen atoms (H).
What type of chemical bond holds ca2+ and o2 together in Cao?
Explanation: Because the bond forms between a metal (Ca) and a non-metal (O), the bond will most likely be ionic.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
