where are the periods on the periodic table, check these out | Where are the periods and groups on a periodic table?
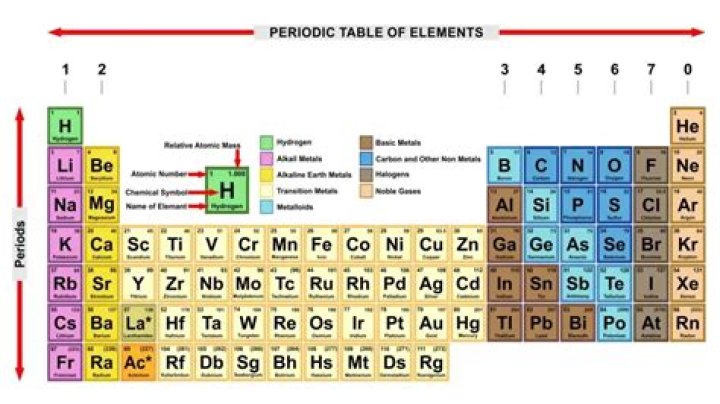
The horizontal rows of the periodic table are called periods. Each period corresponds to the successive occupation of the orbitals in a valence shell of the atom, with the long periods corresponding to the occupation of the orbitals of a d subshell.
Where are the periods and groups on a periodic table?
A period is a horizontal row of the periodic table. A group is a vertical row of the periodic table.
What are the seven periods in periodic table?
There are seven periods for naturally occurring elements:
Period 1: H, He (does not follow the octet rule)Period 2: Li, Be, B, C, N, O, F, Ne (involves s and p orbitals)Period 3: Na, Mg, Al, Si, P, S, Cl, Ar (all have at least 1 stable isotope)
What is a period number on the periodic table?
The horizontal rows on the periodic table are called periods, where each period number indicates the number of orbitals for the elements in that row, according to Los Alamos National Laboratory.
How many periods are in the periodic table?
The periodic table of the elements contains all of the chemical elements that have been discovered or made; they are arranged, in the order of their atomic numbers, in seven horizontal periods, with the lanthanoids (lanthanum, 57, to lutetium, 71) and the actinoids (actinium, 89, to lawrencium, 103) indicated
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
