Which best describes ionization energy Edgenuity?
ionization energy, also called ionization potential, in chemistry and physics, the amount of energy required to remove an electron from an isolated atom or molecule.
Which best describes ionization energy Edgenuity?
Which best describes ionization energy? The ionization energy increases because the ratio of the protons to electrons increases.
What is ionization energy quizlet?
What is ionization energy? The energy required to remove one electron from each atom in one mole of gaseous atoms under standard conditions.
Which of the following is the best definition of first ionization energy?
By definition, the first ionization energy of an element is the energy needed to remove the outermost, or highest energy, electron from a neutral atom in the gas phase.
What are the characteristics of ionization energy?
It is considered a measure of the tendency of an atom or ion to surrender an electron or the strength of the electron binding. The greater the ionization energy, the more difficult it is to remove an electron. The ionization energy may be an indicator of the reactivity of an element.
Which best explains why ionization energy tends to decrease?
The ionization energy decreases because the full s orbital shields the electron entering the p orbital. Which best explains why ionization energy tends to decrease from the top to the bottom of a group? Electrons get farther from the nucleus.
Which of these elements has the highest ionization energy?
Thus, helium has the largest first ionization energy, while francium has one of the lowest.
What is ionization energy what is first ionization energy quizlet?
First ionization energy is the energy required to pull an electron off a neutral Atom. Second ionization energy is the energy required to pull off an electron off of a plus one charge Adam.
What does the second ionization energy refer to quizlet?
The energy required to remove the _____________ electron is called the first ionization energy. The energy to remove the second electron is called the second ionization energy and the energy required to remove the third electron is called the third ionization energy.
Why does ionization energy increase quizlet?
Why does Ionization Energy increase up? There are less shells so Protons are closer to Electrons and less shielding. This means the positive pull is felt more so the electrons are held on better.
Which of the following best describes how ionization energy changes down a group?
What is ionization energy? Which of the following best describes how ionization energy changes down a group? Ionization energy decreases because electrons are closer and require more energy to remove.
What is ionization energy class 11?
Ionization energy. Ionization energy. It is the amount of energy required to remove electron from valence shell of isolated gaseous atom. The word required is used because it means ionization energy is positive that is it means it is always given from outside to remove electron.
What is sodium ionization energy?
This energy is usually expressed in kJ/mol, or the amount of energy it takes for all the atoms in a mole to lose one electron each. For instance, the ionization energy of Sodium (alkali metal) is 496KJ/mol (1) whereas Chlorine’s first ionization energy is 1251.1 KJ/mol (2).
What is ionization energy describe its trend in the periodic table?
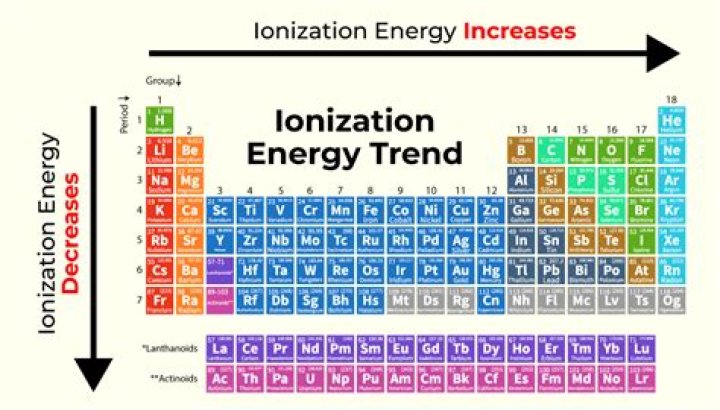
Ionization energy refers to the amount of energy needed to remove an electron from an atom. Ionization energy decreases as we go down a group. Ionization energy increases from left to right across the periodic table.
What is the unit of ionization energy?
In physics, ionization energy is usually expressed in electronvolts (eV) or joules (J). In chemistry, it is expressed as the energy to ionize a mole of atoms or molecules, usually as kilojoules per mole (kJ/mol) or kilocalories per mole (kcal/mol).
Which of the following explains why ionization energy decreases down a group?
On the periodic table, first ionization energy generally decreases as you move down a group. This is because the outermost electron is, on average, farther from the nucleus, meaning it is held less tightly and requires less energy to remove.
Which explains the change in ionization energy that occurs between removing the first and second electrons from an atom?
Which explains the change in ionization energy that occurs between removing the first and second electrons from an atom? The ionization energy increases because the ratio of the protons to electrons increases.
How do ionic bonds affect the properties of ionic compounds quizlet?
How do ionic bonds affect the properties of ionic compounds? The bonds prevent ions from moving throughout the crystal, so a solid ionic compound is a poor conductor.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
