Which of the complex shows ionization isomerism?
(4) Br-andSO-4 can exchange their positions between coordination sphere and ionisation sphere. Hence it shows ionization isomerism.
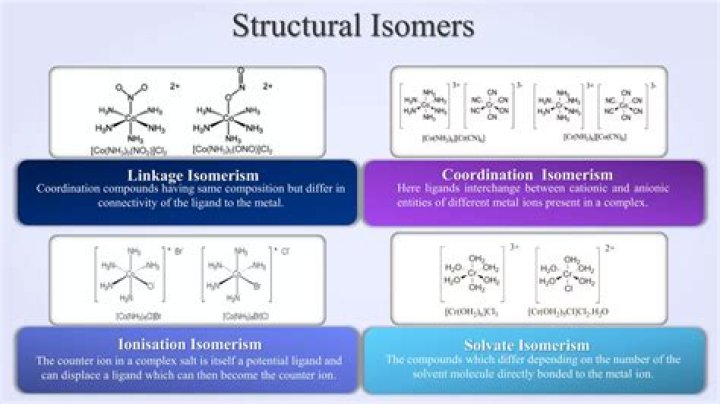
What is an example of ionization isomerism?
Ionisation isomerism is seen to occur in a complex salt when its counter ion is a potential ligand and this counter ion can displace a ligand making it to behave or become a counter ion. > One example of ionisation isomerism is [Co(NH3)5SO4]Br and [Co(NH3)5Br]SO4.
How do you find ionization isomers?
Ionization isomers are identical except for a ligand has exchanged places with an anion or neutral molecule that was originally outside the coordination complex. The central ion and the other ligands are identical. For example, an octahedral isomer will have five ligands that are identical, but the sixth will differ.
Which of the following complex is an outer orbital complex?
Tris(ethane-1, 2-diamine)nickel(II) chloride is homoleptic outer orbital complex.
What are ionization isomers give an example shaala?
Isomers that involve the exchange of ligands between coordination and ionization spheres are called ionization isomers.
Which one of the following will show geometrical isomerism?
But-2-ene will show geometrical isomerism.
Is FeF6 3 an outer orbital complex?
[Fe(CN)6]3– is an inner orbital complex whereas [FeF6]3– is an outer orbital complex.
What is outer orbital complex with example?
In this case, are involved in hybridization and form octahedral complexes. Such a complex in which the central metal ion utilizes outer nd- is called outer-orbital complex. configuration of metal ion on complexation . Consequently, all the octahedral complexes of Co2+,Ni2+,Cu2+and Zn2+ions are always outer-orbital.
Which of the following is an outer orbital complex fecn6?
Hybridisation is d2sp3. In [Fe(H2O)6]2+ is an outer orbital complex while [Fe(CN)6]4- is an inner orbital complex.
What are fac and mer isomers?
Fac/Mer isomers arise when there are two equal types of ligand in an octahedral metal complex. • Fac – When one type of ligand occupies. one face of the octahedron. • Mer – Each set of identical ligands. occupy a plane of the octahedron (2 of the same ligands are trans to each other, the other ligand is cis to these.)
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
