general
Why Calcium chloride is ionic?
Written by Marcus Reynolds — 0 Views
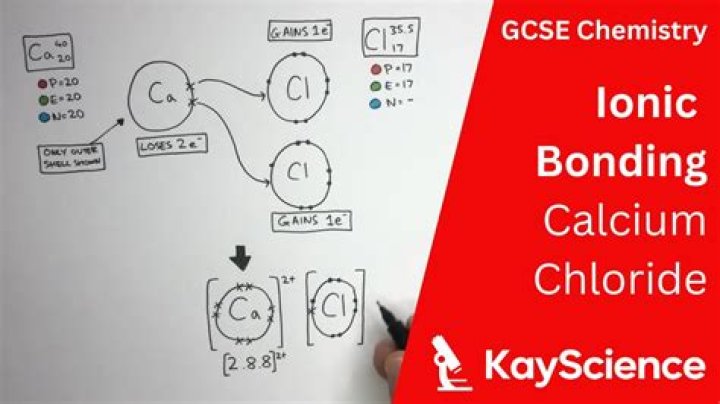
CaCl2 is an ionic compound owing to the large electronegativity difference between the calcium atom and chlorine atom, which is greater than 2.0. In calcium chloride, the calcium atom donates its two electrons and become cation whereas each chlorine atom gain one electron, donated by Calcium, and get a negative charge.
Related Archive

harry potter wizards unite wand guide, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter villain test, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uk edition books, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
