Why does boron only need 6 electrons?
Two of them are core electrons and the remaining 3 are valence electrons. The valence electrons may participate in bonding through sharing with other atoms, to make three bonds. Three bonds = six electrons. That is the simple answer.
Why does boron only want 6 electrons?
The problem with this structure is that boron has an incomplete octet; it only has six electrons around it. Hydrogen atoms can naturally only have only 2 electrons in their outermost shell (their version of an octet), and as such there are no spare electrons to form a double bond with boron.
Can boron have 6 electrons?
Notice that the central boron atom has only 6 electrons in the final Lewis diagram/structure of this molecule. We might conclude from this one example that boron atoms obey a sextet rule. However, boron will form a stable ion with hydrogen, BH4–, in which the boron atom does have a complete octet.
The boron atom has only six electrons in its outer shell, leading to an electron deficiency. This molecule has 12 valence shell electrons; 3 each from the B atoms, and 1 each from the six H atoms.
Does boron gain or lose electrons?
Boron atomic number 5 has five electrons in its ground state. Commonly Boron will lose 3 electrons leaving 2 electrons in its most common ionic form.
In some compounds, the number of electrons surrounding the central atom in a stable molecule is fewer than eight. Since beryllium only has two valence electrons, it does not typically attain an octet through sharing of electrons.
Is boron reactive or stable?
Boron is a poor conductor of electricity, and is fairly non-reactive, although it is water soluble.
Why is boron electron deficient?
Why boron forms electron deficient compounds? Boron has only three electrons in the outermost shell which it can share with other atoms. Hence in their compounds, there are only six electrons present around B-atom i.e. octet is not complete. Hence, boron forms electron deficient compounds.
This means that the number of electrons in the element is equal to the number of protons in the element. The charges of the protons and neutrons will balance out, thus meaning that the element has a no charge. Boron has 5 protons and only 4 electrons which means that it has a positive charge (the charge is +1).
Which element has 6 valence electrons and 3 energy levels?
OXYGEN FAMILY: All have 6 VALENCE ELECTRONS (electrons in the outer energy level) Oxygen, sulfur, and selenium are NONMETALS.
What element has 5 electron shells?
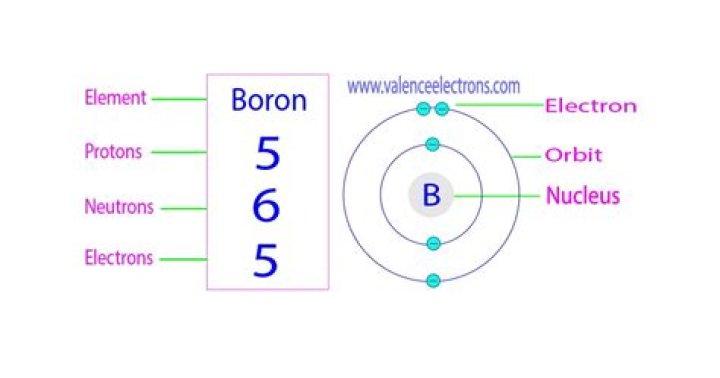
So for the element of BORON, you already know that the atomic number tells you the number of electrons. That means there are 5 electrons in an boron atom. Looking at the picture, you can see there are two electrons in shell one and three more in shell two.
How many electrons and protons does boron have?
Each boron atom has five protons in its nucleus (atomic core), and five electrons. More: Boron is a naturally occurring element. In the environment, boron is combined with oxygen and other elements in compounds called borates.
Boron has 5 electrons — 2 in the first shell, and 3 in the second shell (so three valence electrons). Carbon has 6 electrons — 2 in the first shell, and 4 in the second shell (so four valence electrons). Nitrogen has 7 electrons — 2 in the first shell, and 5 in the second shell (so five valence electrons).
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
