Why is if5 square pyramidal?
IF5, the Iodine atom has 7 valence electrons in molecular orbitals. Thus, Iodine will form 5 bonds with 5 Cl atoms using 5 electrons from its molecular orbital, two electrons will form one lone pair on Iodine atom, which gives the square pyramidal geometry to IF5 molecule.
Is IF5 trigonal pyramidal?
In IF5 there is a lone pair of electrons on iodine where as in PF5 there are no lone pairs on phosphorus. As a result, the molecular shape of IF5 is square pyramidal and PF5 is trigonal bipyramidal.
How is a square pyramidal geometry formed?
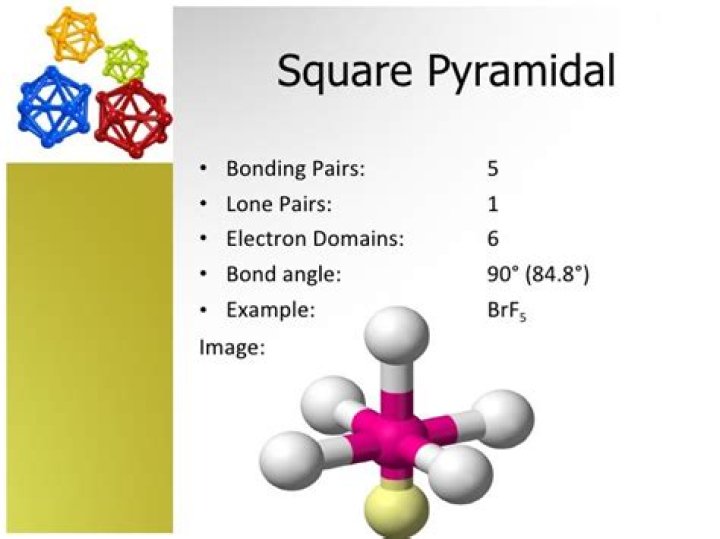
Square pyramidal is a molecular shape that results when there are five bonds and one lone pair on the central atom in the molecule. Bromine pentafluoride (BrF5) has the geometry of a square pyramid, with fluorine atoms occupying five vertices, one of which is above the plane of the other four.
IF5 has a bent square Pyramidal shape because of lone pair and bond pair repulsion.
Which molecule has trigonal planar?
Structure of boron trifluoride, an example of a molecule with trigonal planar geometry.
In IF5, considering I to be the central atom, it has 7 valence electrons. 5 of them form Sigma covalent bonds with 5,F atoms. However this contains a lone pair of e which are not counted in the structure or geometry. There for this forms a Pyramidal structure with a lone pair.
How is trigonal pyramidal difference from square pyramidal?
Trigonal bipyramidal coordination has angles of 90, 120 and 180°. Square pyramidal has no 120° angles, and the 180° angles might be somewhat reduced.
What is difference between square pyramidal and octahedral?
Square planar is a molecular shape that results when there are four bonds and two lone pairs on the central atom in the molecule. The shape of the orbitals is octahedral. Two orbitals contain lone pairs of electrons on opposite sides of the central atom.
Central atom will at the apex and three other atoms or ligands will be at one base, where they are in the three corners of a triangle. There is one lone pair of electrons in the central atom. So when the position of the lone pair is neglected, the remaining bonds make the trigonal pyramidal geometry.
Which of the following has pyramidal shape?
PCl3 has sp3-hybridised phosphorus, with one lone pair. Therefore, molecule has pyramidal shape like ammonia.
Is square pyramidal polar?
With one lone pair on this electronic geometry, the molecular geometry is now square pyramid. POLARITY: NON-POLAR – As stated above, the whole molecule has a perfect symmetry and therefore non-polar (assuming equivalent atoms in the peripheral positions).
Is if5 polar?
IF5 is polar in nature. The molecule has a bent shaped geometrical structure because of lone pair and bond pair repulsion as per VSEPR theory due to which there occurs an imbalance in charge distribution across the molecule.
Bond angles also contribute to the shape of a molecule. The bond angle can help differentiate between linear, trigonal planar, tetraheral, trigonal-bipyramidal, and octahedral. The ideal bond angles are the angles that demonstrate the maximum angle where it would minimize repulsion, thus verifying the VSEPR theory.
What does trigonal pyramidal look like?
Trigonal pyramidal is a molecular shape that results when there are three bonds and one lone pair on the central atom in the molecule. Molecules with an tetrahedral electron pair geometries have sp3 hybridization at the central atom. Ammonia (NH3) is a trigonal pyramidal molecule.
Why is phosgene trigonal planar?
All the atoms present in the molecule are in a plane with a triangular shape, therefore the geometry designated as “Trigonal Planar”. Phosgene (COCl2) is another example that can be taken to represent this geometry. Molecules where the three atoms are not identified such as H2CO deviate from this idealized geometry.
What is square planar molecular geometry in chemistry?
In square planar molecular geometry, a central atom is surrounded by constituent atoms, which form the corners of a square on the same plane. The square planar geometry is prevalent for transition metal complexes with d8 configuration.
Trigonal planar: triangular and in one plane, with bond angles of 120°. Tetrahedral: four bonds on one central atom with bond angles of 109.5°.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
