why is iron used in the haber process, check these out | Why is iron used in ammonia Haber process?
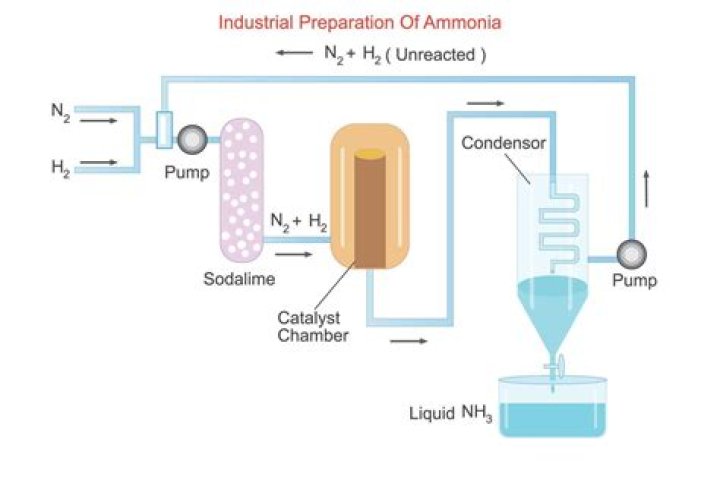
An iron catalyst is used in the Haber process because iron is durable, cheap and effective at catalysis . Catalysts are substances which can speed up the rate at which a reaction takes place. The Haber process is an industrial process in which hydrogen and nitrogen serve as raw materials for the production of ammonia.
Why is iron used in ammonia Haber process?
Iron is a cheap catalyst used in the Haber process. It helps to achieve an acceptable yield in an acceptable time. State three reaction conditions that are controlled in industrial reactions.
Why is iron a good catalyst?
Transition metals are good metal catalysts because they easily lend and take electrons from other molecules. A catalyst is a chemical substance that, when added to a chemical reaction, does not affect the thermodynamics of a reaction but increases the rate of reaction.
What is the function of iron in the reaction of ammonia?
Iron is a biological catalyst that is used in several processes. Iron maintains the equilibrium at different concentrations in the reaction.
How can iron be used as an industrial catalyst?
Abundant iron oxide harnessed to help metals convert carbon dioxide into useful products would simultaneously reduce emissions and add value to waste streams. Current methods to prepare metal-oxide catalysts, the workhorses of chemical transformations, require high temperatures and pressures.
Is iron a transition metal?
The most abundant transition metal in Earth’s solid crust is iron, which is fourth among all elements and second (to aluminum) among metals in crustal abundance.
Why iron and sulphur are heated to produce a compound?
When iron filings and sulphur powder are mixed and heated they undergo a chemical reaction and form ferrous sulphide (FeS). It is a new substance which has properties entirely different from Fe and S. Therefore, heating of a mixture of iron and sulphur powder is a chemical change.
Why iron is not a catalyst?
It is well noted that iron has many limitations as a catalyst for organometallic, organic reactions. It is indeed not electron dense enough to withstand chemical reactions while maintaining its ligand attachments.
Why is iron catalyst used as fine powder?
Answer: fine powder particles provide higher surface area for the catalyst to work efficiently. whereas large prices provide less surface area.
How is iron used?
Most of it is used to make steel — an alloy of iron and carbon — which is in turn used in manufacturing and civil engineering, for instance, to make reinforced concrete. It is used in kitchen cutlery, appliances and cookware such as stainless steel pans and skillets.
What is the importance of iron in modern context?
In modern society iron is the most important of all metals, as it is used to craft different types of steel which is used in a diverse array of applications. Steel is used to make paperclips, skyscrapers, and everything in between. Iron is also an important element in plant and animal life.
Why is iron the most stable element?
The reason iron has a stable nucleus is because of its large nuclear binding energy. Nuclear binding energy is the force that holds the nucleons (protons and neutrons) together in the nucleus of an atom.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
