Why is N2O linear?
Due to two sigma bonds and zero lone pairs around center atom (nitrogen), shape of N2O molecule is linear.
Is N2O a linear structure?
The molecular geometry of N2O is linear.
It has two surrounding atoms and no lone pair. So we can see it has linear molecular geometry and linear electron geometry as well.
Why N2O is linear but polar?
We know that $ C{O_2} $ is a linear and symmetrical molecule so the net dipole moment is zero. Hence, it is non polar. Now, $ {N_2}O $ is also linear and symmetrical but it is considered as the resonance hybrid of two structures due to which its net dipole moment is $ 0.116D $ . Hence, it is polar.
NO2 is a bent molecule; however, when you remove an electron from it, making it NO2+, the molecule becomes linear due to the loss of a lone electron. On the other hand, nitrogen dioxide, NO2, is an AX2E species, and it has an angle of 134 degrees. The additional lone pair on the SF2 molecule makes the angle smaller.
Is nitrous oxide linear molecule?
Nitrous oxide is a linear molecule, so the statement that it is not a linear molecule is not true. The nitrous oxide on inhalation causes an intoxicating effect, so it is true that it is known as laughing gas.
What type of bond is nitrous oxide? All gases are nitrogen and oxygen. Therefore, the most common type of bonding will be covalent.
Does N2O have resonance structures?
Dinitrogen monooxide, or N2O , has three resonance structures, out of which one is a major contributor and one is a minor contributor. Since oxygen is more electronegative than nitrogen, the negative charge will be stabilized better by the oxygen atom.
Why is N2O more polar?
JEE Question
Polarity arises from a difference in electronegativity. If we consider NO2 and N2O both, there is some electronegativity difference. As we know that NO2 is bent structure whereas N2O is linear, so the dipole moment is more in NO2 and hence it is more polar.
N2O is a neutral molecule, nitrous oxide, it is isoelectronic with CO2. The intermolecular force which polar molecules take part in are dipole-dipole forces.
Which is more polar CO2 or N20 give reason?
N2O is more polar as compared to CO2. CO2 is a non polar molecule as the resultant of the two dipole forces is equal and opposite to the third force. This indicates that the dipole moment of the molecule becomes zero.
Why NO2 is bent and CO2 is linear?
Why are NO2 and CO2 different shapes? two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2. a greater repulsion than the single electron in NO2, so the O-N-O angle is reduced further, to 115.4°.
Is NO2 a structure?
Nitrogen Dioxide (NO2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. At room temperatures, nitrogen dioxide is a reddish-brown gas that has a density of 1.8 g/dm3.
Nitrous oxide (N2O) is called “laughing gas” because it has a euphoric effect when inhaled. It has a mildly sweet odor and taste. N2O was first synthesized by J. Priestley in 1772.
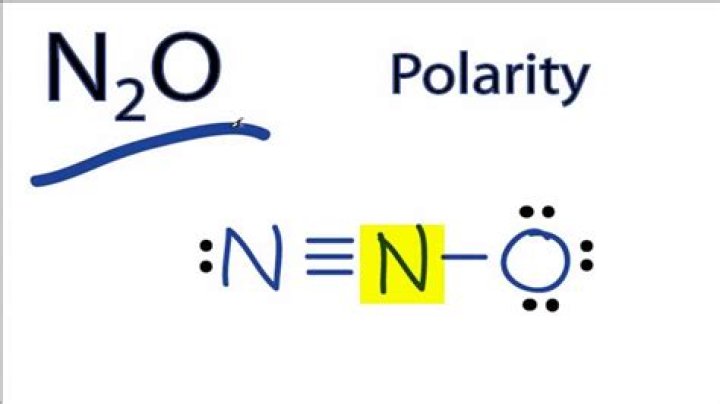
Is N2O a polar bond?
The Lewis structure for N2O is: The central N is bonded to different elements, N and O, making it polar. Furthermore, the N–N triple bond has no dipole moment (ΔEN = 3.04 – 3.04 = 0.0) while the N–O bond has one (ΔEN = 3.44 – 3.04 = 0.40). This results in a net dipole in the molecule.
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
