Why is SiO2 a covalent bond?
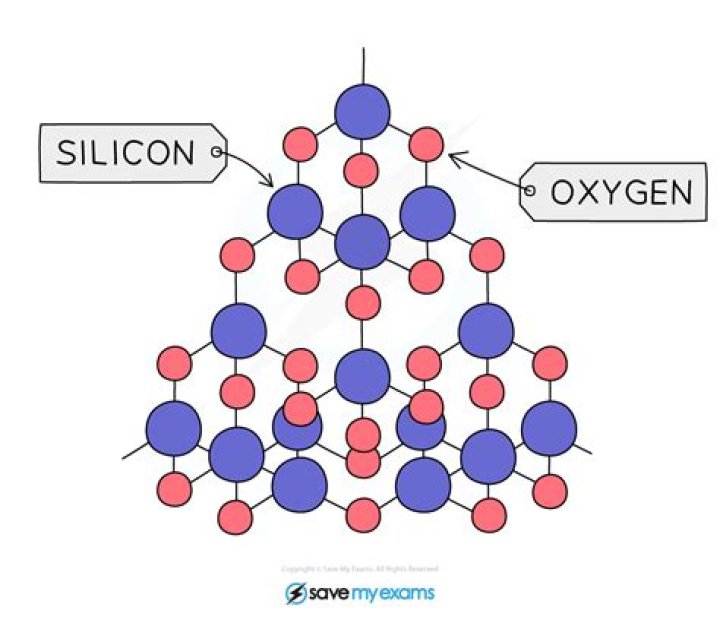
Silicon dioxide is a covalent compound, rather it is a “polar covalent compound”, in which each silicon atom of a crystal is covalently bonded with four atoms of oxygen and each oxygen is bonded with two silicon atoms. … Hence, silica behaves like a covalent compound.
Why is SiO2 covalent?
Silicon dioxide (often called silica) is the main compound found in sand. It contains many silicon and oxygen atoms. All the atoms in its structure are linked to each other by strong covalent bonds. The atoms are joined to each other in a regular arrangement, forming a giant covalent structure.
Why does silicon make covalent bonds?
Silicon (Si) is tetravalent in nature just like Carbon (C) . That means it can easily share all four of its valence electrons to form covalent bonds with other atoms or molecules. So in order to be stable, Silicon(Si)needs to form four covalent bonds. Like Carbon (C), it can form covalent bonds with Hydrocarbons.
Silicon Dioxide (Silica)
Each silicon atom is covalently bonded to four oxygen atoms. Each oxygen atom is covalently bonded to two silicon atoms. This means that, overall, the ratio is two oxygen atoms to each silicon atom, giving the formula SiO2.
Is SiO2 molecular or covalent?
Silicon dioxide, quartz, is a network solid that is covalently bonded. The bonds are covalent because the electronegativities of Si and O are 1.9 and
Silicon dioxide is also known as silica or silicon(IV) oxide has three different crystal forms. Very strong silicon-oxygen covalent bonds have to be broken throughout the structure before melting occurs. Morevoer, it hard due to the need to break the very strong covalent bonds.
What is covalent bond in silicon?
The outermost shell of atom is capable to hold up to eight electrons. Silicon atom forms four covalent bonds with the four neighboring atoms. In covalent bonding each valence electron is shared by two atoms.
How does silicon bond to other silicon atoms?
In the silicon crystals that form the backbone of the electronics industry, each silicon atom forms covalent bonds with four other silicon atoms, sharing one of its electrons (and receiving a shared electron in return) from each of the four neighbors.
With its four valence electrons, silicon can form covalent or ionic bonds either donating or sharing its four shell electrons. At the same time, it is a relatively inert element and does not react with oxygen or water in its solid form.
Is SiO2 polar or nonpolar covalent bond?
And that the Lewis structure of Silicon dioxide does not have any lone pairs of electrons. So there is no shape distortion, making it easy to cancel out the dipole moment produced by the Si-O bond. As a result of all of these factors, SiO2 is a non-polar molecule.
Is SiO2 a molecule or compound?
The chemical compound silica, also known as silicon dioxide, is known for its hardness since the 16th century. It is found in nature in many different forms, such as flint, quartz, and opal. Silica (quartz): Silica, SiO2, is a chemical compound that is composed of one silicon atom and two oxygen atoms.
Is SiO2 a macromolecule?
Silicon dioxide, or silica, (SiO2) is another important example of a macromolecular solid. Silica can exist in six different crystalline forms. The best known of these is quartz, whose crystal structure shown previously is shown again below.
SiO2 has a linear shape, and since the elements at each end are the same, the pull is canceled out, making the overall compound non-polar.
Is sulfur dioxide ionic or covalent?
Sulfur dioxide is a Covalent compound because, in the case of sulfur dioxide, two atoms possessing similar electronegativity try to bond together. In doing so, the slight difference of electronegativity between the two atoms results in sharing of electron bonds forming covalent bonds.
Why is SiO2 not a molecule?
Silicon dioxide is not formed of small molecules. It consists of an infinite array of silicons where each silicon is bonded to four separate oxygens (and each oxygen is shared between two silicons). This creates a strong refractory solid (glass and sand are mostly silicon dioxide aka silica).
Is SiO2 ionic molecular metallic or covalent?
In other words, anything that only uses ionic or covalent bonding will have high melting points, high boiling points and be relatively hard and rigid. There are two common examples of such systems. Quartz, or SiO2, is composed exclusively of covalent bonds. Table salt, or NaCl, is composed exclusively of ionic bonds.
This means that SiO, is only moderately ionic and the character of its chemical bond niay be considered as being between the covalent (fi = 0) and the ionic limit (fi = 1).
Related Archive

harry potter wizards unite apple, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform shop, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter wand name list, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
