Why NH4OH is not a strong electrolyte?
answer:NH4OH is a weak electrolyte with dissociation constant equaled to 1.76 10-5. It dissociates partially into ions in an aqueous solution. …
Why NH4OH is not a strong electrolyte?
The ammonium hydroxide dissociates to generate a hydroxide ion which recombines with the ammonium ion to produce ammonium hydroxide. It does not undergo $text{ 100}{scriptstyle{}^{0}/{}_{0}}text{ }$ dissociation. Thus ammonium hydroxide is a weak electrolyte.
Is NH4OH a weak electrolyte?
answer:NH4OH is a weak electrolyte with dissociation constant equaled to 1.76 10-5. It dissociates partially into ions in an aqueous solution.
Is NH4OH the strongest electrolyte?
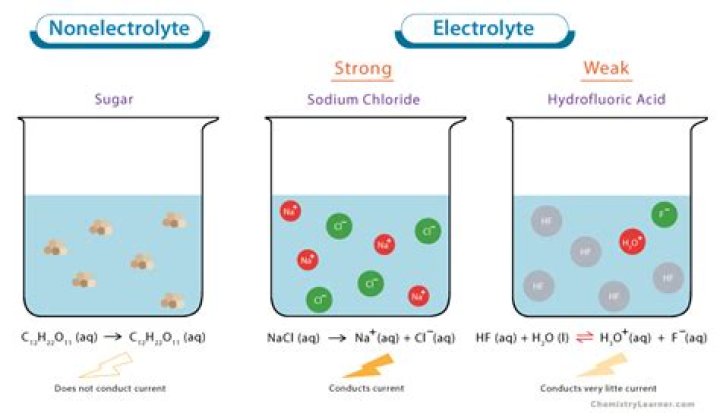
Ammonia, NH3(aq), or ammonium hydroxide, NH4OH(aq), is a weak base and therefore a weak electrolyte.
Is NH4OH strong or weak?
NH4OH a weak base because it has higher H+ concentration than a stronger base. Ammonia is a typical weak base. Ammonia itself obviously doesn’t contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions.
Why is NH4OH considered a weak base?
Ammonium hydroxide (NH4OH) is a weak base because the production of the number of OH– ions are very low as compared to the number of NH3 we dissolved in water, this is because ammonia is itself a weak base, and it’s only a tiny fraction of it reacts with water to form(NH4+ and OH–), hence, an only small fraction of OH–
Is ch3ch2oh an electrolyte?
It is a non-electrolyte that does not form ions in water.
Is HNO2 strong or weak?
Nitrous acid (HNO2), a weak acid, is very unstable and exists only in aqueous solution.
Is ch3coona a strong electrolyte?
CH3COONa is a salt of weak acid and strong base. Since sodium acetate is a salt therefore it will dissociate completely in water and hence acts as a strong electrolyte.
What substances are strong electrolytes?
Strong Electrolytes
hydrochloric acid, HCl.hydroiodic acid, HI.hydrobromic acid, HBr.nitric acid, HNO3sulfuric acid, H2SO4chloric acid, HClO3perchloric acid, HClO4
Which of the following is strong electrolyte *?
A strong electrolyte is a solute or solution that is an electrolyte that completely dissociates in a solution. HCl (hydrochloric acid), H2SO4, NaOH and KOH are all strong electrolytes.
Is RbOH a strong base?
(a) RbOH is a strong base. A weak base is a chemical base that does not ionize fully in an aqueous solution. In chemistry, a base is a substance that can accept hydrogen ions (protons) or, more generally, donate a pair of valence electrons. The base dissociation constant (Kb) measures a base’s relative strength.
Why NaOH is a strong and NH4OH is weak electrolyte?
Sodium hydroxide (NaOH) is strong base because it fully dissociates in water to produce hydroxide ions. While ammonia (NH3) is weak base because it accepts protons from water to produce fewer hydroxide ions in solution. While weak bases produce fewer hydroxide ions, making the solution less basic.
Is acetic acid a strong electrolyte?
We therefore make a distinction between strong electrolytes, such as sodium chloride, and acetic acid, which is an example of a weak electrolyte. As the name acetic acid suggests, this substance is also an acid, as well as a weak electrolyte. Accordingly, we classify acetic acid as a weak acid.
Related Archive

harry potter trivia show host, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter uniform pattern, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023

harry potter vans ebay, latest free online harry potter movies, best HD videos you should watch in 2022 – 2023
